Scientists at IBM Research in the US have shown that nanotubes made of vanadium oxide are magnetic at room temperature. Moreover, the magnetic properties of the nanotubes can be controlled by doping them with electrons or holes. The work could have applications in spintronic devices that exploit the spin of the electron as well as its charge to perform logic operations (L Krusin-Elbaum et al. 2004 Nature 431 672).
Like their carbon counterparts, nanotubes made of vanadium oxide display a variety of electronic properties depending on their detailed structure. Lia Krusin-Elbaum and co-workers at IBM’s TJ Watson Research Center in New York made multiwalled vanadium oxide nanotubes with a self-assembly technique that relied on organic molecules called amines to direct the growth of the nanotubes. The tubes produced were typically several microns long with diameters between 60 to 100 nanometres.
There were three types of vanadium atom in the structure: so-called V(2) and V(3) sites are low energy sites with relatively localized electrons which have spins that point in opposite directions. This gives rise to an overall ferromagnetic moment because there are two V(2) spins for every (V3) spin. However, the entire nanotube is non-magnetic because the itinerant electrons associated with the higher energy V(1) sites “frustrate” the magnetic order by making it impossible to form a unique ordered ground state.
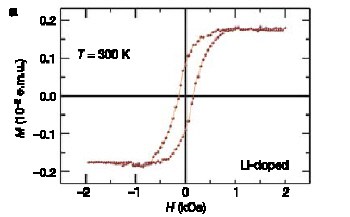
Krusin-Elbaum and colleagues found that when they doped their nanotubes with extra electrons — which they did by adding lithium — the structures became magnetic (see figure). Even more surprising, the nanotubes also became magnetic when the addition of iodine removed electrons to create holes.
“Removing electrons takes out enough of the V(1) electrons to eliminate their roles as spins, while lithium doping adds enough electrons to approach double occupation of the V(1) sites, which also renders them spinless,” Krusin-Elbaum told PhysicsWeb. “Hence the net result is that either kind of doping turns on ferromagnetism, in a system whose undoped state is non-magnetic.”
The team now plans to develop nanosystems in which the electron spins can be controlled by a voltage or some other physical mechanism, rather than doping.




