Researchers in the US have developed a new microscopy technique that can pinpoint unlabelled molecules in biological tissue at depths of up to several millimetres. This is much deeper than current methods, which are limited to about 100 µm. Called vibrational photoacoustic (VPA) microscopy, the technique has been used to make 3D images of plaque lining arteries and could be used for diagnosing diseases such as atherosclerosis.
In recent years, scientists have developed microscopy techniques that can locate specific molecules in a biological sample without the need to label those molecules. Although techniques such as stimulated Raman scattering and coherent anti-Stokes Raman scattering have revolutionized biological imaging, their use is limited by their relatively small penetration depth.
Now, a team led by Ji-Xin Cheng at Purdue University has increased this depth by being the first to demonstrate VPA microscopy. Exploiting the photoacoustic effect in imaging and microscopy is not a new idea, but what the researchers do differently is to use the effect to target specific molecules.
Picking up vibrations
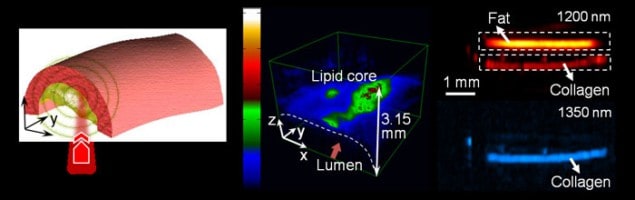
The technique involves firing a laser pulse at a sample to excite a specific vibrational mode associated with the carbon–hydrogen bonds that abound in body fat. The wavelength of the pulse is chosen so that absorption by blood and surrounding tissue is minimal. The laser pulses cause the fat molecules to heat and expand locally, thus generating pressure waves at ultrasound frequencies that are detected by a transducer. By scanning the laser over the sample in 2D and measuring the arrival time and intensity of the ultrasound at a number of different locations, the team is able to create a 3D image giving the location of fat in the sample.
“Targeting specific chemical bonds is expected to open a completely new direction for the field,” says Cheng. “Measuring the time delay between the laser and the ultrasound waves gives a precise distance, which enables you to image layers of tissue and create 3D pictures using just one scan.”
To demonstrate the potential of 3D VPA imaging, carotid arteries were removed from pigs with profound atherosclerosis. The team detected a strong VPA signal from fat molecules located 1.5 mm below the illuminated surface of the sample, allowing the identification of different levels of fat accumulation. The VPA technique clearly distinguished a number of different fatty deposits in the arteries. This is important in the study and diagnosis of cardiovascular diseases because fat combines with other substances to form artery-clogging plaque. The researchers also used VPA microscopy to map the distribution of fats in fruit-fly larvae.
Next step is miniaturization
The Purdue group is now looking to miniaturize its system and develop a catheter-based imaging device. “We are hoping to build an endoscope to put into blood vessels,” says Cheng. “This would enable us to see the exact nature of plaque formation in the walls of arteries and to better quantify and diagnose cardiovascular disease.”
Team member Han-Wei Wang adds that the spatial resolution of the VPA system is suitable for such future work. “The lateral resolution is very flexible from the order of a micrometre to tens of micrometres,” he says. “The resolution is an improvement compared with current clinical imaging methods such as intravascular ultrasound. Our spatial resolution will be enough for atherosclerotic applications, and will be a great option as a complementary imaging modality.”
Although the first area of interest for the Cheng group is cardiovascular disease, in the future the method might also be used to detect fat molecules in muscles to diagnose diabetes or other lipid-related disorders, including neurological conditions and brain trauma. The technique can also image protein fibrils, making it useful when studying collagen’s role in scar formation.
The work is described in Physical Review Letters.