Understanding how the body’s immune system responds to viral infections is essential for developing new vaccines and improved treatments. The recent pandemic reiterated this need, prompting particular interest in the role of T cell response to COVID-19 infection.
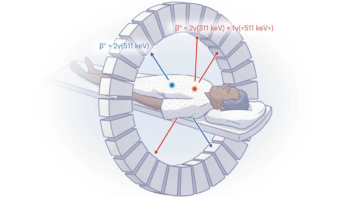
Previous studies of human immune response relied on peripheral blood assays; but most T cells reside in tissue rather than blood. What’s needed is a non-invasive way to quantify immune cell biodistribution and kinetics in vivo, particularly in lymphoid organs. With this aim, researchers at UC Davis used the uEXPLORER total-body PET scanner to perform first-in-human immunoPET imaging of CD8+ T cell biodistribution in three healthy individuals and five patients recovering from COVID-19. They report their findings in Science Advances.

The team used an immunoPET imaging probe based on a 89Zr-labelled minibody (an antibody fragment) with high affinity to human CD8 (expressed on the surface of CD8+ T cells). The long half-life of 89Zr (78.4 h) allows the radiotracer’s biodistribution to be followed for several days after injection. However, 89Zr-based immunoPET imaging with conventional clinical PET scanners suffers from high noise and concerns over radiation dose.
“The high statistical noise was the major barrier for dynamic imaging and kinetic modelling of immunoPET tracers with conventional PET scanners,” explains first author Negar Omidvari. “Total-body PET scanners have extended axial length compared to conventional PET scanners, resulting in a significant increase in sensitivity. We expected that total-body PET would bring significant improvements in imaging 89Zr-based tracers, while at the same time allowing for dose reduction and total-body dynamic imaging.”
T cell tracking
The eight participants in the study received low doses (mean activity of 18.8 MBq) of radiotracer via intravenous infusion. The researchers then performed three total-body PET/CT scans: a 90-min dynamic PET scan starting immediately before the infusion, plus two 60-min PET scans 6 and 48 h later. No adverse effects or changes in participants’ vital signs were observed during the study.
Despite a sixfold reduction in radiation dose, the total-body PET scans exhibited significantly better image quality than previous human PET scans with this radiotracer. In particular, a large number of high-contrast lymph nodes were visualized in all subjects. Standardized uptake value (SUV) images at the three imaging timepoints showed high tracer uptake in lymphoid organs in all subjects, with the highest uptake observed in the spleen. Tracer uptake in peripheral lymph nodes was seen in all subjects as early as 30–90 min post-infusion, peaking at 48 h.
Time–activity curves for all organs-of-interest revealed consistent trends in tracer kinetics, with SUVs in bone marrow regions increasing during the dynamic scans in all subjects and plateauing towards 90 min. Between the 6 and 48 h timepoints, SUV decreased in the spleen, bone marrow and lungs, and increased in the lymph nodes and tonsils, for all subjects. During the last 42 h, changes in SUV were similar in all organs-of-interest, with no significant differences between COVID-19 patients and controls.
The researchers also calculated tissue-to-blood ratios (TBRs), defined as the ratio of tissue activity to whole-blood activity. Plotting TBRs as a function of time revealed distinct differences between COVID-19 patients and controls in all bone marrow regions at early timepoints (up to 7 h). The team note that subject 2 (who had been infected with COVID-19 twice) had TBRs of all bone marrow regions, spleen and tonsils two to three times higher in than all other patients.
These changes were not evident in the SUV images, suggesting that TBR values are more informative for assessing CD8 distribution.
“Tissue SUVs are directly related to concentrations of the radiotracer in blood, which change as a function of time,” Omidvari explains. “Therefore, if there are differences in blood clearance of the radiotracer between different patients, comparing tissue SUVs between patients at a specific timepoint becomes prone to error. Also, when imaging with immunoPET tracers, T cell trafficking can affect the SUVs at later timepoints, because T cells can get labelled and then move to a different tissue.”
Better than bloods
For comparison, the researchers performed peripheral blood assays on samples drawn from each subject before radiotracer infusion. Flow cytometry revealed higher percentages of CD8+ T cells, activated CD8+ T cells and CD8+ memory T cells in the peripheral blood of COVID-19 convalescent patients compared with controls.
The COVID-19 patients, who were first scanned within eight weeks of symptom onset, also underwent a second set of PET/CT scans and blood assays roughly four months later. Comparing baseline with 4-month scans revealed increased TBRs in all bone marrow regions of all subjects. The researchers note that, although this study included a small number of subjects, the immunoPET platform appears more sensitive than blood assays for studying CD8+ T cell physiology in individuals.
Ultrasensitive PET enables sub-second total-body imaging
“We see a huge difference in the PET data of subject 2 in many different tissue types at all timepoints, which is not visible in the blood data,” says Omidvari. “We also see a small increase in bone marrow TBRs of all COVID-19 patients at 4-month follow-up scans compared to their baseline scans. However, in peripheral blood CD8 assay, we don’t see these differences or patterns.”
The team concludes that dynamic immunoPET imaging is currently the only non-invasive technology that can provide in vivo insight into whole-body T cell distribution and trafficking in human subjects, with the use of total-body PET enabling acceptable radiation dose burden.
“Next, we plan to improve the kinetic modelling to include the T cell trafficking pathway into the model, to not only quantify the trafficking rates in different tissues, but also to decouple the effect of T cell trafficking on tissue uptake,” Omidvari tells Physics World.