Theoretical physicists in Italy and France have discovered a new family of "magic" clusters using computer simulations. The clusters, which consist of a nickel or copper core surrounded by silver atoms, display high levels of structural, thermodynamic and electronic stability. The silver-nickel structures are also magnetic (G Rossi et al. 2004 Phys. Rev. Lett. 93 105503).

Bimetallic nanoclusters are used in applications as diverse as catalysis and optics, and their properties depend on both their size and chemical composition. Clusters that contain a magic number of atoms have advantages for many applications because they are more stable than other clusters.
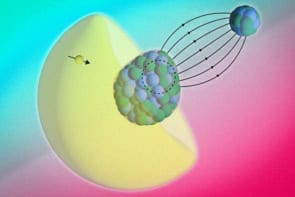
Physicists recently discovered a new type of silver-nickel nanocluster that comprised a well-defined silver shell with a nickel core. Now, Riccardo Ferrando and colleagues in Genova, Milan, Ghezzano, Trieste and Marseille have shown theoretically that there is a whole new family of core-shell polyicosahedra clusters. The polyicosahedra are built by packing together icosahedra that contain 13 atoms as shown in figure 1.
Ferrando and colleagues used a “genetic algorithm” to look for the structures with the lowest energy, and therefore the most stability, among selected magic sizes of clusters. They physicists began with polyicosahedra (pIh) that contained N1 silver atoms, N2 copper or nickel atoms, and m interpenetrating icosahedra. This structure is referred to as (N1, N2) pIhm. They found that for a total of 38 atoms the most stable clusters were (30, 8)pIh8, (31, 7)pIh7 and (32, 6)pIh6. For a cluster with 34 atoms the most stable structure was (27, 7)pIh7 (figure 2).
According to the team, the stability of these clusters arises from the different sizes of the atoms (the silver atoms are much bigger), and the fact that metals like to form short bonds with external atoms and long bonds with internal atoms. The tendency of the silver atoms to segregate at the surface is also important. Ferrando and colleagues now plan to look for similar clusters made from other elements and to investigate their catalytic, optical and magnetic properties.