Researchers in the US have combined tin oxide "nanobelts" with low-power microheaters to make sensors that can detect nerve agents. The devices made by Li Shi and colleagues at the University of Texas at Austin and the Georgia Institute of Technology are ultrastable, highly sensitive and free from the "poisoning effect" that has previously limited the widespread use of metal oxides as sensors (C Yu et al. 2005 Appl. Phys. Lett. 86 063101).

Metal oxide sensors work by measuring the changes in electrical conductance that take place when gas species undergo reduction or oxidation (redox) reactions with the surface of the sensor. The detection sensitivity increases as the sensors are made thinner, but a process called grain boundary poisoning limits the reliability and long-term stability of thin-film sensors.
Metal oxide sensors require high operating temperatures to enhance redox reactions. Shi and co-workers therefore integrated tin oxide nanobelts — ribbon-like single-crystal structures that are as little as 10 nanometres across — with microfabricated heaters. This method reduces power consumption and means that battery-operated miniaturised sensor arrays can be made.
The Texas-Georgia Tech researchers trapped a nanobelt between a pair of platinum electrodes and then deposited a thin platinum coating on the contacts between the nanobelt and the electrodes (figure 1). They then mounted the sensor in a small flow-through chamber and tested its sensitivity to low concentrations of dimethyl methylphosponate (DMMP), a compound that is often used to simulate nerve agents, and nitrogen dioxide in air.
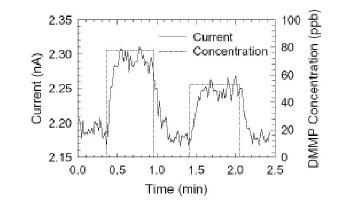
The electrical conductance though the device increased by about 5% when it was exposed to 78 parts per billion (ppb) of DMMP in air at 500°C and changed with the concentration of the nerve agent stimulant (figure 2). Moreover, it was sensitive to concentrations of nitrogen dioxide as low as 200 ppb at 200°C. This is an order of magnitude more sensitive than similar devices.
The current through the nanobelt recovered less than 3 minutes after the gas flow was shut off and the chamber purged with air. In contrast, previous devices took as long as 40 minutes to recover. According to the team, this improvement is due to the absence of grain boundaries in the nanobelt structure and the use of the platinum coating.
“The mass production of devices based on ‘bottom-up’ synthesised nanomaterials has remained a challenging task,” says Shi. “Our work shows that integration of bottom-up synthesised metal oxide nanobelts with ‘top-down’ fabricated microelectromechanical systems (MEMS) can be a viable and efficient approach to large-scale manufacturing of well-organised sensor arrays.”