It is usually a good idea to keep water away from electrical equipment but researchers in Japan have discovered a new effect by breaking this rule. Masahide Gunji and Masao Washizu of the University of Tokyo have shown that electric fields can be used to move water droplets around a solid surface. Their work could lead to new ways to perform chemistry experiments much faster than is possible at present (J. Phys. D38 2417).
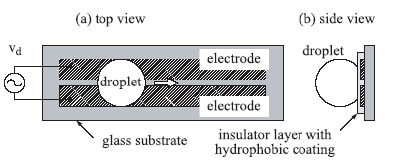
The Japanese team began by making a pair of parallel-strip electrodes on a glass substrate that had been covered by a thin electrically insulating hydrophobic layer and then placing a water droplet on the surface (figure 1). The droplet, which had a volume of between about 1 nanolitre and 1 microlitre, adopted an almost spherical shape due to a combination of surface tension and wetting effects.
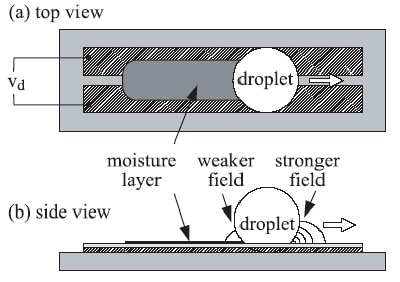
To their surprise, the team found that the droplet began moving at speeds of up to 10 centimetres per second when a voltage was applied across the electrodes (figure 2). The initial direction of motion was unpredictable and the droplet continued to move until it reached the edge of the device or the voltage was switched off. Moreover, when the electrodes were made into the shape of a racetrack, the droplet kept travelling around the track (figure 3).
This motion came as a surprise because the electrodes used in the experiment create a uniform electric field along their length, so there is no energy gradient to move the droplets. According to Gunji and Washizu, the motion is “self-propelling” (figure 4). “The droplet leaves behind a moisture layer on the substrate surface, which shields the trailing edge of the droplet from the electric field,” says Washizu. “This produces an imbalance in the electrostatic force exerted on the droplet, which, in turn, provides the driving force for the droplet to move.”
It might be possible to use the new effect to automate chemistry experiments. “Many chemistry experiments are labour-intensive and are performed by mixing chemicals in test-tubes,” says Washizu. “If small liquid droplets could be transported across a substrate then chemical reactions could, in principle, be induced in small volumes by simply colliding the droplets together and mixing their contents.”