Physicists have made a new type of molecule in a gas of ultracold atoms confined to one dimension. The molecules can form when the interaction between two atoms is either attractive or repulsive, which is completely different to what happens in free space (Phys. Rev. Lett. 94 210401). The result could help test a variety of many-body theories in quantum physics and may even provide insights into the nature of high-temperature superconductivity.

In three dimensions atoms only bind together to form molecules when the scattering length that describes the interactions between them is positive. Now, Tilman Esslinger and colleagues at ETH Zurich in Switzerland have found that if the atoms are confined to move in just one dimension, then weakly bound molecules can be created for both positive and negative scattering lengths.
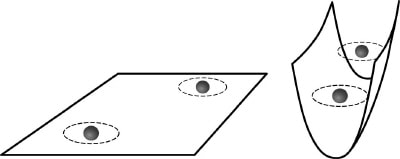
The ETH team began by loading an ultracold gas of potassium-40 atoms into an optical lattice made from crossed laser beams. The pattern created by the interfering laser beams resembles an array of “tubes” in which the particles can only move back and forth along the axis of the tube (figure 1). The gas was so cold that the potassium atoms – which have to obey the Pauli exclusion principle because they are fermions – occupied all the available low-lying quantum states to create a degenerate Fermi gas.
Next, Esslinger and colleagues used a magnetic field to change the scattering length by exploiting a Feshbach resonance. As first predicted by Maxim Olshanii of Harvard University in 1998, the atoms form molecules for both positive and negative scattering lengths because they are unable to pass each other freely in the tube (figure 2). The ETH team confirmed that the binding energy of the molecules agreed with theoretical predictions by using a radio-frequency field to split them apart.
“Our experiment provides an excellent testing ground for quantum many-body theories,” says Michael Köhl. “For example, exact theories are available for the cross-over between a superfluid BCS state in which the atoms form pairs (the same as in a superconductor) and a Bose-Einstein condensate of molecules. .”
In addition to possibly shedding new light on the electron-pairing mechanism in high-temperature superconductivity, the one-dimensional atomic gas created by the ETH team could also be used to explore the properties of Luttinger liquids – one-dimensional systems in which the interactions between particles are much more important than in three dimensions.