Silver atoms become magnetic when grouped together in small clusters, according to new computer simulations by physicists in Spain. The result is surprising because silver atoms are normally diamagnetic in the bulk. If made for real in the lab, magnetic silver clusters could be used for a variety of biomedical applications (physics/0605160).
Clusters of atoms and ions form a type of matter that is intermediate between single atoms and bulk matter. Metallic clusters are widely used as catalysts because they have a very high surface to volume ratio, which allows them to speed up chemical reactions. Researchers have, however, recently begun to see if magnetic clusters can be used in biomedicine — for example, to separate labelled biological cells, to improve drug delivery and to enhance contrast in magnetic resonance imaging.
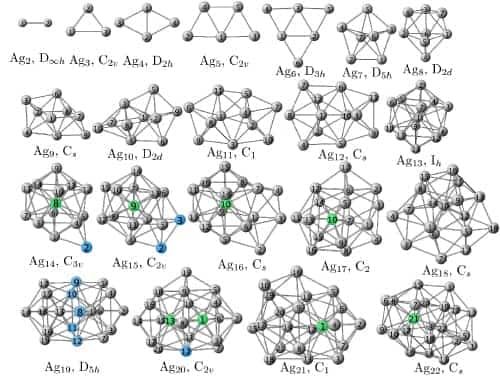
The new study, carried out by Manuel Pereiro and colleagues at the University of Santiago de Compostela, involved performing “density functional” calculations using an off-the-shelf computer package. The calculations involved solving the Schrödinger equation for groups of atoms arranged into a cluster and searching for sliver clusters with the lowest energy and hence the highest stability. To do this, the researchers analysed a huge sample of trial geometries, containing between 2 and 22 silver atoms (figure 1). Of these clusters, they then looked at those structures that had the highest magnetic moment.
Pereiro and co-workers found that the most stable cluster with the highest magnetic moment contained 13 silver atoms (figure 2). According to the team, this is because the cluster has a highly symmetric icosahedral symmetry. Symmetry allows the silver atomic orbitals to become degenerate, or have the same energy, which, in turn, produces magnetism. Clusters bigger than 13 atoms have a lower magnetic moment per atom because they have distorted icosahedral symmetry; smaller clusters have a lower magnetic moment due to their different, unstable, shapes.
According to the researchers, the silver-13 cluster has a high magnetism because atoms at the edge of the cluster transfer electrons to the atom in the middle — to make this inner atom more energetically stable. The charge transfer reduces the inner atom’s magnetism and boosts that of the outside atom. This is because the number of outer atoms with partially filled orbitals (that is, unpaired spins) increases while the number of inner atoms with unpaired spins decreases. (Only atoms with unpaired spins can exhibit magnetism in the absence of an external magnetic field). Overall, this leads to an increase in the average magnetic moment of the Ag13 cluster.
Such clusters could be used in medicine because they are more biocompatible and less toxic than conventional metallic clusters, which make them ideal for therapeutic drug delivery applications. Confirming the magnetic properties of clusters in the lab would also be “a golden opportunity for experimentalists”, says Pereiro.





