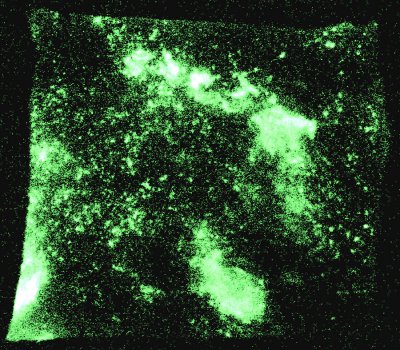
Watch somebody munch on boiled sweets in the dark, and you might just see their mouth glow. That's because piezoelectric materials such as sugar can emit light when they are broken. Now researchers at the University of Illinois in the US have made this "mechanoluminescence" up to 1000 times brighter by pummelling crystals with ultrasonic shock waves (Nature 444 163).
In materials with structures that lack symmetry such as piezoelectric crystals, charge of opposite sign separates when the material is stressed. If the stress is great enough, the material fractures, and the charge then recombines in the gas between the gap producing a small spark of light. Kenneth Suslick and Nathan Eddingaas from Illinois found that they can intensify the effect by immersing slurries of crystals into paraffin infused with various gases, which is then irradiated with ultrasonic waves.
During this process, millions of microscopic bubbles continually form and implode through the process of “acoustic cavitation”, generating shock waves that drive individual crystals together at half the speed of sound. “It’s like the difference between hitting the crystals with a hammer and firing at them with a bullet,” said Suslick.
The team monitored the light emitted from two known piezoelectric crystals: sugar and resorcinol (a white aromatic solid). When they were manually crushed in nitrogen, the crystals not only displayed luminescence, but also a faint nitrogen emission spectrum. Both types of crystals produced the same emission when they were irradiated with ultrasound in nitrogen-infused paraffin – however, the intensity was so much greater that the crystal slurries could be seen glowing in daylight. Moreover, only irradiation with ultrasound could produce spectra from paraffin infused with inert gases such as helium or argon.
Suslick and Eddingaas said that the bolstered intensity was because of the increased rate of fracture from the 20 kHz ultrasonic waves, which meant more surfaces were exhibiting mechanoluminescence at any one time. But only the strength of the collisions could explain the presence of line emissions for helium or argon, which are much more difficult to ionise. “We learn more about the fracture of materials from this kind of study,” said Suslick, who has been investigating acoustic cavitation for the past 30 years. “It’s a tool for understanding chemistry at the interface between the mechanical world and the molecular world.”