What do stampeding elephants and flocks of birds have in common with living cells from a goldfish? They can all be described by a new model of "flocking" devised by biophysicists in Hungary. The model is based on observations of live cells that begin to move in unison as their numbers reach a critical level. It is the first to explain how such simple organisms can do this without being "aware" of their neighbour's collective motion. (Phys. Rev. E to be published).

Just as some birds fly in unison in large flocks, some bacteria and other live single cells can organize themselves into moving structures. But exactly how this happens has been a mystery because unlike birds, single cells have no conceivable way of observing and responding to the average velocity of their neighbours.
Now Bálint Szabó and colleagues at Eötvös University in Budapest have developed a new theory of flocking that is unique because it is not based on individuals being aware of their neighbour’s average velocity. The researchers say that this very simple model of flocking could also be applied to a wide variety of animals – even complex organisms such as birds or even elephants.
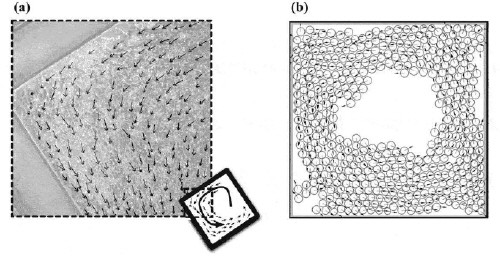
The researchers studied the collective motion of keratocytes, which are live single cells produced by goldfish scales. Fish keratocytes are often used in studies of cell migration because they move rapidly and their movement is not affected by the presence of chemicals in their environment.
The cells were confined within a small incubator and their behaviour was recorded visually using a videomicroscope (see figure “Fish cells on the move”). When only a few cells were present, the keratocytes moved independently in random directions. However, as the population multiplied, the keratocytes started to move collectively when the cell density reached about 0.0005 cells per square micrometre. Above this critical density the keratocytes moved in coherent groups. When the cell density was increased even further, the effect of collisions with the walls of the square-shaped incubator caused the cells to move in a whirl-like structure (see figure “Whirling keratocytes”).
The researchers were able to explain this phase transition by creating a simple model of the interaction between two keratocytes that is based on three forces acting at three different distances. At very short separations a repulsive force causes the keratocytes to move apart. At intermediate cell separations (up to about one cell diameter) an attractive force causes the keratocytes to move together. At distances greater than about one cell diameter the force was set to zero. When used to simulate the behaviour of moving cells, this combination of simple forces caused the onset of collective motion at a critical density. This is unlike previous models, which assumed that the cells could respond to the motion of their neighbours.
The team hopes their experiment and model will create a better understanding of a range of biological phenomena, including how groups of cells of arrange themselves to become embryos and how wounds heal by the coherent movement of endothelial cells.