When you are trying to get from A to B, friction is both friend and foe. Friction between tyres and the road allows a car to be steered, for example, whereas axle friction reduces the vehicle’s fuel efficiency. Now, however, biophysicists in Germany have discovered that friction also plays an important role in how molecular motors move within living cells.
The discovery could lead to a better understanding of cell biology because molecular motors are involved in a wide range of processes, such as DNA replication and sperm mobility.
The new study has been carried out by Joe Howard and colleagues at the Max Planck Institute of Molecular Cell Biology and Genetics, along with Erik Shaeffer at the Technical University of Dresden, who looked at “kinesin” molecules. These comprise a large family of molecular motors that transport cargoes (such as biological molecules) along microtubular strands.
Molecular motors get legless
Biophysicists believe that kinesin molecules have two “legs” and walk along the microtubule much like a human on a tightrope — gripping the microtubule with one leg, while releasing the other and pulling it forward. But because the molecules are only a few nanometres in size, thermal fluctuations make them more like drunks walking along a rapidly shaken tightrope.
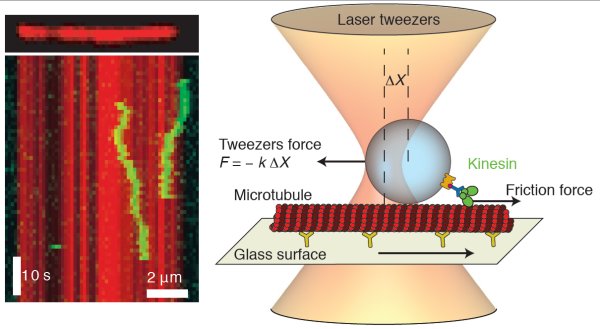
To understand the forces involved in keeping the motors on track, the team attached kinesin to a tiny bead about 1 μm in diameter. They then released the bead into the focal point of a laser beam, with the optical pressure on the bead holding it firmly in place in “optical tweezers”. A microtubule mounted on a translation stage was then positioned next to the bead and pulled along at a range of speeds of about 1 μm/s, which is roughly the natural speed of the molecular motor.
The team then determined the frictional force on the ball by measuring the deflection of the ball from the centre of the tweezers. At speeds below 1 μm/s, the friction was proportional to the relative speed of the kinesin and the microtubule. This was confirmed in a different experiment by observing the motion of kinesin that had run out of fuel (ATP). These molecules diffuse along microtubules at a rate defined by Einstein’s diffusion equation, which can also be used to calculate friction.
Taking it in their stride
When the relative speed was above 1 μm/s, friction still increased — but the rate of increase began to fall. According to Howard, this is exactly what should happen if kinesin’s legs are grabbing onto specific sites on the microtubule and then letting go. By analysing the shape of the force/speed curve, the team concluded that the “stride” of the motors is about 8 nm, which also happens to be the length of a certain dimer that repeats along the microtubule. This came as a surprise to the team, because previous attempts at measuring this stride using other techniques suggested 4 nm.
Howard told physicsworld.com that the results “give us a new way of thinking about how molecule motors operate — that two elements are involved, force generation and friction”. This could help biophysicists understand how large numbers of motors work together to propel a sperm cell, for example, because, says Howard, such collective motion is likely to involve friction.
The studies could also help scientists understand motion on a much larger scale. According to Shaeffer, the forces that stick the kinesin legs to the microtubule are the same Van der Waals forces that allow some geckos scurry up a smooth wall. Geckos rely on tiny strands on their feet that are about 100 nm in size, whereas kinesin legs use structures about 25 nm in size.
The work is reported in are reported in the 14 August edition of Science.