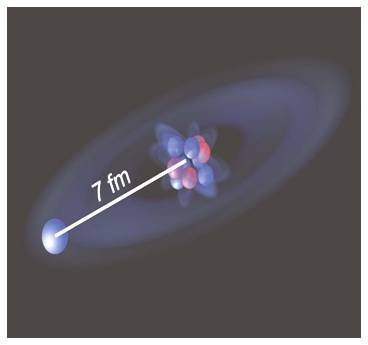
Physicists in Europe and North America have measured the radius of an unusual beryllium isotope containing a single neutron a long way from the rest of the nuclear core. Although the radii of other such “halo” isotopes have been determined before, this is the first time that the measurement has been made on a nucleus with just a single halo neutron. The researchers found that the halo neutron in beryllium–11 is, on average, about 7 fm (7 x 10-15 m ) from the nuclear core, which itself has a radius of about 2.5 fm.
First discovered in 1985, halo nuclei have a conventional nuclear core plus one or more halo neutrons that spend much of their time a relatively long distance away. The lithium–11 halo nucleus, for example, has about the same diameter as the much more massive uranium nucleus. The reason such nuclei are so large is that the energy that binds halo neutrons to the core is only 100 keV — roughly a tenth of the energy tying neutrons in a conventional nucleus.
But measuring the size of halo nuclei has proven tricky because the nuclei are very short lived — and because they have no electrical charge, the halo neutrons do not interact readily with experimental probes.
Blur of positive charge
The best measurements have involved studying the tiny “volume” shift — of about one part in a billion — of the energy levels of the electrons that are bound to a halo nucleus in an atom or ion. This shift occurs because the halo neutron and the core orbit each other and their relative motion makes the core appear as a blur of positive charge to the electrons. This, for example, means that the electron energy levels of a beryllium atom containing the halo nucleus beryllium–11 are shifted slightly compared to atoms containing the more conventional nuclei beryllium–7, beryllium–9 or beryllium–10.
The volume shift can then be used to calculate the radius of the blur of positive charge, which can then be used to calculate the average separation between the halo and core.
While physicists have already managed to measure this shift in helium and lithium halo isotopes, experiments on beryllium–11 nuclei are further complicated because beryllium has four electrons.
It turns out that electron energy levels are also affected by “mass shifts” that are caused, in part, by interactions between the nucleus and the correlated motion of the electrons. These mass shifts are about 1000 times larger than the volume shift and become increasingly difficult to calculate as the number of electrons increases. The problem is simplified somewhat by studying Be+ ions, which only has three electrons.
Measurements and calculations
The new study was carried out at the ISOLDE facility at CERN by Wilfried Nörtershäuser at the University of Mainz and colleagues in Germany, Canada and Switzerland (Phys. Rev. Lett. 102 062503).
The experiment involved producing four different isotopes of beryllium (with 7, 9 10 and 11 nucleons) by firing a 1.4 GeV proton beam into a uranium-carbide target. This created beryllium atoms, which were then ionized using a laser and accelerated to 50 kV. Transitions in electron energy levels were induced by firing two ultraviolet laser beams at the ions. One beam was fired straight at the oncoming ions, while the other was fired in the opposite direction from behind the ions to cancel out the experimental uncertainty in the kinetic energy of the ions.
Some of the laser light is absorbed by the beryllium’s electrons, which jump to a higher energy level. As the electrons fall back down, they emit light at the same wavelength as the laser through the process of “resonance fluorescence”. However, the wavelength of the light absorbed and then emitted by the halo isotope beryllium–11 differs very slightly from the light emitted from conventional beryllium isotopes — the difference being due to the isotope shift, which is the sum of the mass and volume shifts.
The team determined this tiny shift by using a device called a frequency comb, which is capable of making a very accurate measurement of the laser’s wavelength. By comparing the resonant wavelengths of beryllium–11 with the other beryllium isotopes — and then correcting for the mass shift — the team worked out the volume shift. This allowed them to conclude that the halo neutron is about 7 fm from the nuclear core. The core itself has a radius of about 2.5 fm.
Improving mathematical models
“The halo neutron is thus much farther from the other nucleons than would be permissible according to the effective range of strong nuclear forces in the classical model”, explained Nörtershäuser. “The result can now be used by nuclear physicists to improve their mathematical models of nuclei”, he said
Jim Al-Khalili at the University of Surrey in the UK told physicsworld.com, “These measurements tell us quite clearly that the core of the halo nucleus beryllium–11 (namely, beryllium–10) is more tightly packed together than a much lighter nucleus like beryllium–7.” He added. “We learn a lot about the core within the halo with this work, and indirectly we can test our theoretical models of how the halo particles interact with the core”.



