Physicists in the US have created an ultracold gas of molecules with “adjustable” dipole moments. The experiment, which is the first to study the effect of long-range dipole interactions in an ultracold gas, could lead to new ways of using trapped molecules to simulate quantum effects that occur in solids
Ultracold gases make ideal “quantum simulators” because some of the interactions between the component atoms or molecules can be “tuned” by adjusting the applied magnetic and laser fields that keep the particles in place. While physicists have been successful at dialling up short-range interactions between atoms and molecules, simulating long-range interactions – such as those between charged particles – has proven more difficult.
Earlier this year Jun Ye and colleagues at the National Institute of Standards and Technology (NIST) in Colorado and Maryland cooled potassium-rubidium (KRb) molecules to see how they react chemically to form Rb2 and K2. By doing so, they were able to observe how the initial quantum states of the molecules affect reaction rates – something that cannot be observed at room temperature.
Now, the same NIST team has turned its attention to the long-range electric-dipole interaction between the molecules – and how it affects the reaction rate. As in their previous experiment, the team created ultracold KRb molecules by cooling a mixture of potassium and rubidium atoms to just a few hundred nanokelvin and then exposing them to a magnetic field gradient. This binds the atoms together, with the bond being further strengthened by exposing the atoms to laser light.
Direction matters
If there is no applied electric field, the molecules have no electric dipole moment. A pair of molecules will therefore only react if they can tunnel through an energy barrier that arises because the particles are fermions, having non-integer spin. The result is a relatively low reaction rate.
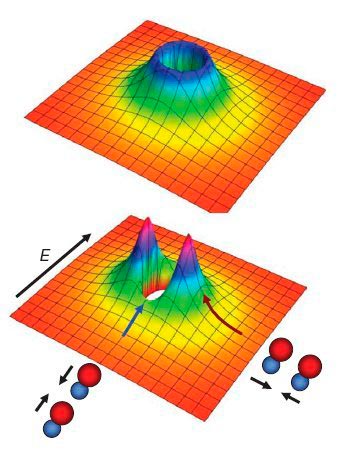
But if a small electric field is applied to the gas, the molecules acquire electric dipole moments that all point in the same direction along the field. So when two molecules collide, they feel a dipole-dipole interaction that depends on the relative orientation of the collision and the electric field.
If the collision occurs along the direction of field, the positive end of one dipole collides with the negative end of the other (a head-to-tail collision) and the force is attractive. However, if the collision is perpendicular to the field, the force is repulsive.
The attractive force lowers the height of the tunnelling barrier, making it more likely that the molecules react. Even though the tunnelling barrier gets bigger for perpendicular collisions, the overall effect is to make the reaction go faster, which should lead to the gas warming up.
The temperature of the gas can be measured by switching off the magnetic trap and determining the rate at which the gas expands – the faster the rate the higher the temperature. By measuring the expansion rate in different directions, the team found that collisions are more likely to occur in a certain direction.
Expanding gas
Ye and colleagues studied the dipole interaction by repeating the expansion measurements for a series of samples that were exposed to different electric fields of different strengths. They found that the reaction rate did not change significantly as the dipole moment was increased – until it reached a specific level, above which the reaction rate increased rapidly.
The results could help physicists to gain a better understanding of how to create long-lived ultracold dipolar gases. According to Ye, the importance of the head-to-tail interactions suggests that the lifetime of such gases could be boosted if they are confined to a 2D “pancake” so that head-to-tail collisions cannot occur. He said the team have already managed to suppress losses due to the dipole-dipole interaction.