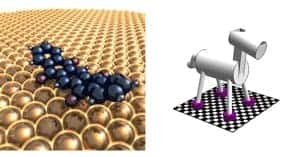
Molecular machines could be capable of quantum mechanical tunnelling – something normally seen in very tiny particles like electrons and atoms. That’s the claim of researchers at the University of California, Riverside who have made and studied “two-legged” and “four-legged” nanomachines.
The tunnelling behaviour has never before been seen in devices so big and is a fundamental departure from mechanics in the macroscopic world, says team leader Ludwig Bartels. It also means that such machines could move much faster than expected.
Molecular machines are found everywhere in biology. For example, the acid in our stomachs is produced by a proton pump in cells that line the stomach. And in every cell in the body, proteins are dragged to the place where they are needed using kinesin motors. These biological motors consist of thousands of atoms and are much too big to be studied using computer models.
Bartels’ team wanted to understand the basic principles behind natural molecular machines so that could develop similar, artificial devices. The researchers, together with chemist Michael Marsella, made small, easy-to-study, molecules that can “walk” and carry “cargo” when placed on a flat copper surface held in vacuum. “This is a very much more simplified set-up than in biology, where molecules need to attach themselves everywhere in 3D and where all kinds of other molecules are floating by,” explains Bartels.
Molecules walk the line
Two years ago, Bartels and colleagues found that anthraquinone (a very common molecule, tons of which are used in the pulp industry) could walk across copper surfaces in a straight line. This was an important result in itself because normal molecules tend to move around randomly in all directions. Moreover, anthraquinone can attach carbon dioxide molecules to its two oxygen atoms, or “legs”, and drag this cargo along too while it walks. However, the researchers were puzzled as to why the molecule moved so fast.
The researchers then studied pentacenetetrone – another widely available molecule – which has not two oxygen “legs” as in anthraquinone but four. To their surprise, Bartels and co-workers found that this “quadrupedal” molecule, which moves like a pacing horse (both legs on one side of the molecule move together followed next by the two legs on the other side), moved a million times slower than “bipedal” anthraquinone.
According to the Riverside researchers, this huge difference in speed occurs because portions of the bipedal anthraquinone simply tunnel through barriers (like surface roughness or corrugations) in the environment rather than climbing over them. Although the quadrupedal molecule can coordinate its four “hooves” so that it paces forward, it cannot coordinate both legs so that they tunnel through a surface barrier at the same time. This means that the molecule needs to move its oxygen legs in the conventional way – over the barriers.
One foot at a time
“The bipedal molecule is a million times faster because it only needs to move one foot at a time,” Bartels told physicsworld.com. One foot can just start tunnelling forward at any time and make a successful step.
It is like driving on a bumpy road with the wheels of your car going through the bumps rather than over them Ludwig Bartels, University of California, Riverside
“This is a fundamental departure from mechanics in the macroscopic world and greatly speeds up movement,” he added. “It is like driving on a bumpy road with the wheels of your car going through the bumps rather than over them. Quantum mechanics allows such behaviour for very light particles, like electrons and hydrogen atoms but could it also be relevant for big molecules like anthraquinone?”
Artificial molecular machines like these might find applications in microelectronics, for example in data storage, or in medicine for drug delivery. However, real-world devices still may be a way off – perhaps 10 years away, says Bartels.
The team now plans to make longer molecules – “think millipedes rather than horses” – that might be driven with light.
The work is reported in J. Am. Chem. Soc. DOI: 10.1021/ja1027343.