For more than a century scientists have relied on the “ergodic theorem” to explain diffusive processes such as the movement of molecules in a liquid. However, they had not been able to confirm experimentally a central tenet of the theorem – that the average of repeated measurements of the random motion of an individual molecule is the same as the random motion of the entire ensemble of those molecules. Now, however, researchers in Germany have measured both parameters in the same system – making them the first to confirm experimentally that the ergodic theorem applies to diffusion.
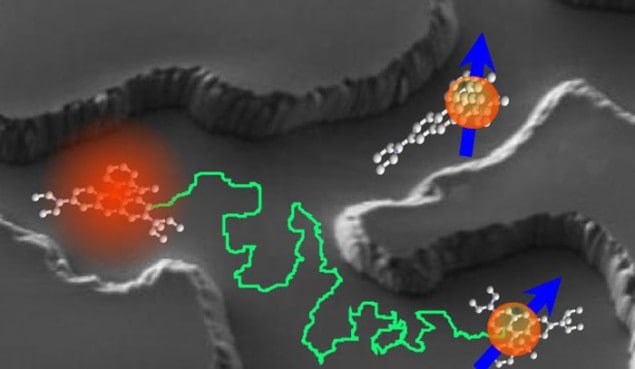
The experiments developed from the work of Christoph Bräuchle and a team at Ludwig-Maximilians University in Munich, who developed a technique for tracking individual dye molecules dissolved in alcohol that then pass through a nanoporous material. Such diffusion is of more than just academic interest because it plays an important role in a number of technologies, including molecular sieves, catalysis and drug delivery.
Pinpoints of light
To confirm the ergodic theorem, Bräuchle’s team tracked the molecules by illuminating the sample with light. This makes the molecules fluoresce so that they appear as pinpoints of light when viewed using a high-powered optical microscope. By using dye molecules at very low concentration, the researchers ensured that each point of light corresponded to just one molecule. So, by measuring the intensity profile of a point and finding its centroid, the Munich team was able to determine the position of a dye molecule to within about 5 nm. Individual molecules could then be followed as they moved through the sample by taking a series of snapshots.
Meanwhile, a team led by Jörg Kärger at the University of Leipzig used a nuclear magnetic resonance (NMR) technique to track the diffusion of all the dye molecules in a similar sample. The pulsed-field-gradient NMR method is sensitive only to the collective motion of all the dye molecules and cannot determine individual molecules. Comparing the results from the two groups showed that the average of many measurements of the diffusivity of individual dye molecules (as measured in Munich) was identical to the collective diffusivity of the dye molecules (as measured in Leipzig). Given that diffusion involves the random motions of molecules, the study therefore confirms the ergodic theorem.
Conflicting requirements
Bräuchle told physicsworld.com that the main challenge was to find a system that could be studied using both techniques. The fluorescence method works best when the dye concentration is extremely low and the molecules move very slowly – whereas the NMR measurements need much higher concentrations and faster motion. The compromise involved using a special microporous material that slowed down the molecules and constrained them to a plane so that they were easier to track with the microscope. In addition, the dye concentration in the NMR experiments was about 10 times greater than that used for the fluorescence measurements.
Now that the researchers have worked out a way to confirm the ergodic theorem, they are keen to use the technique to search for systems that do not obey the theorem. Bräuchle believes that this could occur when some molecules diffuse through living cells – something that could have important implications for how drugs are designed.
The research is described in Angewandte Chemie.