The “Best-in-Physics” session at the AAPM Annual Meeting showcase the studies that score highest in the abstract review process and are judged to reflect the highest level of scientific quality and innovation. The ever-popular electronic poster session includes presentations in therapy, imaging and joint imaging-therapy categories. Here’s our selection from this year’s chosen papers.
Cardiac compensation enhances CBCT scans
“ACROBEAT is all about improving imaging in the interventional cardiac suite, and eventually within radiotherapy,” explained Tess Reynolds from the University of Sydney. Presenting in the joint imaging-therapy category, she described a 3D gated cardiac cone-beam CT (CBCT) protocol that compensates for cardiac rhythm variations during the scan.

Reynolds explains that, currently, cardiac CBCT scans are not adapted for patient motion. The ACROBEAT (adaptive cardiac cone-beam computed tomography) protocol, on the other hand, uses the patient’s electrocardiograph (ECG) signal to regulate the gantry velocity and projection time intervals in real time.
ACROBEAT decreases the number of projections needed by acquiring all images prospectively in a single gantry rotation. This removes the current need for multiple gantry sweeps and eliminates unnecessary projections not used in image reconstruction.
“This improves the image quality, reduces the dose – which is particularly important for cardiovascular imaging – and significantly reduces scan times,” Reynolds told Physics World. Faster scans help reduce the length of the breath hold required. “We can image adaptively in a single sweep, which gives an 84% reduction in imaging dose.”
To date, Reynolds and colleagues have simulated in silico scans. They are now poised to begin experiments on a research C-arm system, using their software to control the gantry.
Another future aim is monitoring the patient’s breathing trace as well as their ECG to perform dual cardiac- and respiratory-motion mitigated imaging (which could eliminate the need for a breath hold). Such an approach opens up the possibility of using ACROBEAT within radiotherapy, for example to enable targeting of central lung tumours by removing the cardiac motion.
Nanoparticles plus radiation boost drug delivery
The delivery of cancer drugs to pancreatic tumours is limited by the presence of intact blood vessels that restrict the permeation and penetration of chemotherapeutics or large molecular drugs. Presenting in the therapy category, Sijumon Kunjachan, an instructor in radiation oncology at Harvard Medical School described an innovative technique to address this problem.

“We are trying to improve drug delivery in pancreatic tumours, by modulating the tumour blood vessels using combined radiation and nanoparticle therapy,” he explained.
The approach is based on the use of gold nanoparticles functionalized with peptides that have an affinity to receptors on tumour endothelial cells. When injected intravenously, the nanoparticles bind to the surface of the tumour blood vessel. During irradiation, these nanoparticles cause the blood vessels to rupture and enable the cancer (nano)drugs to leak through the altered vessels and accumulate in the tumour.
Kunjachan and colleagues tested these nanoparticles in mice bearing a human pancreatic cancer. They injected the functionalized nanoparticles 24 hours prior to external-beam radiation therapy and tested the concept using both preclinical and clinical radiation beams. To determine whether vessel disruption increases permeability, they tested two model drugs: a short-circulating MRI contrast agent and a long-circulating fluorescent contrast.
“We saw that the accumulation of both the agents in pancreatic tumours almost doubled,” said Kunjachan. “The next step will be to test this approach using routinely administered chemotherapeutics or nanodrugs in an orthotopic and genetically engineered pancreatic or lung tumour model, and to observe the tumour regression and survival benefits.”
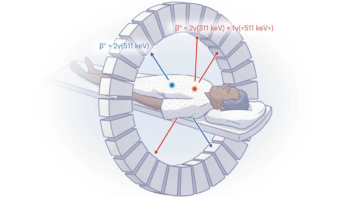
Selenium ramps TOF-PET performance
In the imaging category, Andrew LaBella from SUNY Stony Brook University described NEW -HARP, an amorphous selenium photodetector for time-of-flight (TOF) PET. The aim of NEW-HARP (nano-electrode multi-well high-gain avalanche rushing photoconductor) is to achieve a sufficient time resolution to perform TOF-PET in simultaneous PET/MRI.

LaBella explained that to optimize TOF-PET imaging, the goal is to minimize the coincidence time resolution, ideally to below 100 ps. The resulting improvement in signal-to-noise ratio can then be exploited to increase scanning throughput, reduce dose or enhance lesion detection.
“Current photosensors are costly and have low quantum efficiency,” said LaBella. “So we propose the use of selenium instead of silicon. It is more cost-efficient, offers similar timing and higher quantum efficiency.”
LaBella and colleagues fabricated a multi-well solid-state photomultiplier based on amorphous selenium. To speed up the device performance, they used an embedded grid electrode to achieve unipolar time differential charge sensing – a design that improved the timing resolution to around 100 ps.
The key advantages of NEW-HARP, noted LaBella, are the cost-effectiveness of selenium and the possibility of achieving 100 ps timing. “The benchmark for the field is a coincidence timing resolution of 100 ps, if you can do that, you can justify the cost of TOF-PET and everyone will buy into it,” he explained.
The next steps will be to optimize the fabrication process, and then fabricate and test the coincidence timing of a full ring of PET detectors using NEW-HARP as the photomultiplier coupled to LYSO scintillators.