
Innovations in embedded computing, sensors and batteries enabled the introduction of robotic prostheses designed to help restore locomotion to individuals with disabilities. Such prostheses are moved by motors that respond to the user, via signals from built-in movement sensors, for example.
To improve the response of a robotic device to the user’s intent, it can also incorporate neural information from electromyograms (EMG), which record electrical activity from residual limb muscles. With this aim, researchers from Northwestern University are developing a control system for a robotic lower limb prosthesis that incorporates EMG data and operates in real time.
“Any assistive robotic device should seamlessly and automatically respond to the user’s intent. EMG data contain the neural information that makes this possible and have been used to control prosthetic arms for decades; however, it is challenging to measure EMG data from legs,” explained first author John Spanias, who completed the study as a postdoctoral researcher at the Shirley Ryan AbilityLab. “We wanted to develop and test the technology that could incorporate this important neural information into a prosthesis over long-term use.”
In their latest work, Spanias and colleagues evaluated the performance of their technology on individuals with transfemoral amputations (J. Neural. Eng. 15 016015).
Transitioning locomotion mode
State-of-the-art robotic prosthetic legs produce different mechanical responses when the user is walking on level ground, an incline or stairs, but they are currently unable to automatically transition between these modes of locomotion. This could be achieved by using EMG signals, input into a pattern recognition algorithm, to transition the device between locomotion modes (a process known as forward prediction).
As EMG signals will likely also fluctuate daily as the prosthesis is taken on and off, the team developed an adaptive forward prediction algorithm to track such changes. To prevent misclassification of locomotion mode, the algorithm can revert to sole use of mechanical sensor data when large EMG fluctuations are detected. It is then retrained with updated EMG data, determined by categorizing the user’s gait profile and labelling the patterns with the correct mode (backwards estimation).
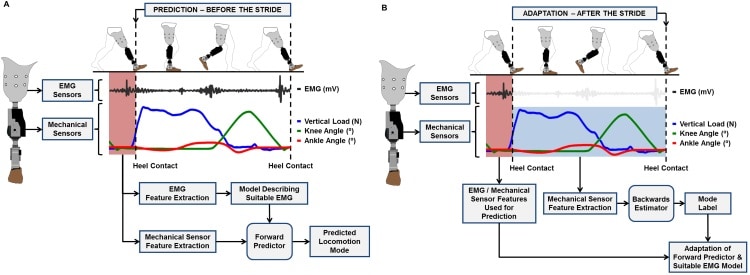
The researchers conducted experiments with eight transfemoral amputees over multiple days, recording eight channels of EMG data and signals from 22 mechanical sensors. In a first experiment, they collected data to train the adaptive forward predictor algorithm while the subjects used the powered prosthesis to walk on level ground, stairs and ramps. During this offline session, the experimenter manually transitioned the prosthesis between modes.
In a second experiment, the subjects completed the same tasks, but mode transitions were automatically controlled by the (previously trained) adaptive forward prediction algorithm. All subjects were able to transition between the required locomotion modes and complete the different activities with low error rates. The adaptive control system accurately responded to the user’s intent over multiple days, despite changing neural signals.
In the first quarter of such a session, EMG was used in 68.86% of forward predictions. This increased to 96.31% by the end of the experiment. The algorithm’s ability to select when to incorporate EMG is important as EMG signals will likely differ across days.
Transitions to and from ramp descent were the most problematic for the forward predictor, and were mostly frequently classified as level walking (8.45%). Such errors typically only presented a minor perturbation to the user. More substantial errors that may endanger the patient, such as inappropriately transitioning into stair ascent while on level ground, were rare (below 1%).
Comparisons between adaptive and non-adaptive algorithms showed that the adaptive system performed significantly better. The overall error rate of the adaptive forward predictor was 4.03%. For non-adaptive systems, the error rates were 4.44% for a system that used only mechanical sensors, and 4.38% for a system based on mechanical sensors and EMG.
Improved control
The researchers also examined the impact of their recent enhancements to the prosthesis control system, which included adding more mechanical sensors to the prosthesis, incorporating a 90 ms delay into the mode transitions and implementing a mode-specific classifier architecture. These modifications decreased the error rate of the forward predictor, with the lowest error rates obtained when applying all modifications. The analysis also revealed that the impact of adding EMG signals was less when these modifications were included.
EMG data do, however, still have an important role to play, says Spanias. “For example, EMG data could still be used in tasks such as moving individual prosthesis joints when the user is not walking, controlling difficult transfers into or out of chairs, or detecting stumbles and falls,” he explained. “Moreover, as our ability to acquire EMG data gets better, we expect that it will help control lots of different ambulation activities.”
As the system learns from the user while they walk, it should improve performance over time. As such, the team is now planning to allow people to use the system at home, to assess its performance over long periods of time. “We are also very excited to apply these algorithms to EMG signals measured from tiny, wirelessly powered implants,” Spanias told medicalphysicsweb. “We expect to be able to extract more information which will result in improved control of prosthetic arms and legs.”



