Particle therapy enables highly conformal radiation delivery while reducing dose to normal tissue. However, the presence of nearby organs-at-risk can limit the maximum achievable tumour dose. One approach that could overcome this limitation is nanoparticle-aided radiotherapy. Researchers from Korea have now investigated the use of Fe3O4/TaOx nanoparticles as dose-enhancing radiosensitizers for proton therapy (Phys. Med. Biol. 63 114001).
Nanoparticles are selectively taken up by tumours, due to the leaky tumour vasculature. Upon irradiation, the nanoparticles emit low-energy electrons that are deposited nearby, thereby enhancing the deposited local dose. The researchers chose the bimetallic nanoparticle Fe3O4/TaOx (core/shell) as it is already used as a contrast agent for CT and MR imaging, thus opening up the potential for theranostic applications.
“Verifying the tumour position with CT or MR images every day during treatment can enhance the accuracy of radiation therapy,” explains Youngyih Han from Samsung Medical Center and Sungkyunkwan University. “In addition, we wanted to investigate whether these bimetallic nanoparticles can enhance the therapeutic ratio as a radiosensitizer.”
In vivo imaging
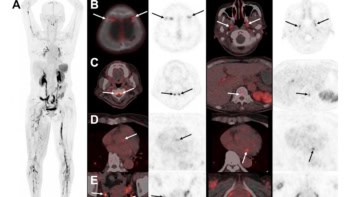
First, Han and colleagues verified the detectability of Fe3O4/TaOx nanoparticles in vivo by imaging six mice bearing mammary carcinomas. They acquired MRI and microCT images of the animal’s organs before, and 5 min, 30 min, 60 min and 24 hr after injection of the nanoparticles in solution.

They observed that the uptake ratio of the tumour region increased with time, with a maximum tumour-to-tissue concentration ratio of 0.16 after 24 hours. In the MR image, the tumour was distinguishable 5 min after nanoparticle injection, and the signal intensity of the tumour region gradually increased with time. In the microCT image, however, the aorta and blood vessels were seen 5 min after injection, but the tumour periphery was not visible until after 24 h. The authors note that improvement in the uptake efficiency is desirable.
Simulation studies
In the second part of this work, the researchers used Monte Carlo simulations to compute the dose enhancement from Fe3O4/TaOx and other nanomaterials. They simulated 70 and 150 MeV proton irradiation of gold, gadolinium, Fe3O4/TaOx, Fe3O4, iodine and BaSO4 nanoparticles located at the centre of a 4x4x4 µm water phantom. They then calculated the dose enhancement ratio (DER) – the ratio of the radiation dose with nanoparticles to the dose without – for each case.
Calculating DER as a function of distance from the nanoparticle surface demonstrated that all nanomaterials had a dose enhancement effect, and that it was greatest near to the particle surface. Irradiation with 70 MeV protons resulted in DERs (at 1 nm) of 15.76, 7.68, 7.82, 6.17, 4.85 and 5.51, for gold, gadolinium, Fe3O4/TaOx, Fe3O4, iodine and BaSO4, respectively. The dose enhancement with 150 MeV proton irradiation was similar.

The researchers expect that the approach will also work for higher proton beam energies, as used in patient treatments. “As shown in the Monte Carlo simulation, the dose enhancement ratios for 70 and 150 MeV were not that different, but we think the dose enhancement could be slightly lower due to the lower yield of secondary electron at higher energies,” says Han.
Han notes that additional in vitro experiments have shown that cells mixed with Fe3O4/TaOx nanoparticles and irradiated with 230 MeV energy proton beams showed a decrease in cell survival compared with a control group without nanoparticles.
For both proton energies, gold generated the largest yield of secondary electrons, followed by gadolinium and Fe3O4/TaOx. The dose enhancement with the Fe3O4/TaOx nanoparticles was approximately half that seen for gold, and similar to that of gadolinium. However, the researchers point out that Fe3O4/TaOx nanoparticles are cheaper to produce than gold nanoparticles, and more biocompatible than gadolinium.
A unique feature of Fe3O4/TaOx nanoparticles is that the Fe3O4 in the core is superparamagnetic. This could enable tumour targeting, by combining this superparamagnetic property with a well-designed magnetic field. “We are investigating active targeting methods for Fe3O4/TaOx nanoparticles to overcome the 50% lower dose enhancement compared with gold,” Han explains.
The authors concluded that Fe3O4/TaOx nanoparticles can be effective cancer cell sensitizers when used with proton therapy. “We are now preparing in vitro and in vivo experiments using proton beams, to link the Monte Carlo simulation results to biological effects,” Han told Physics World.