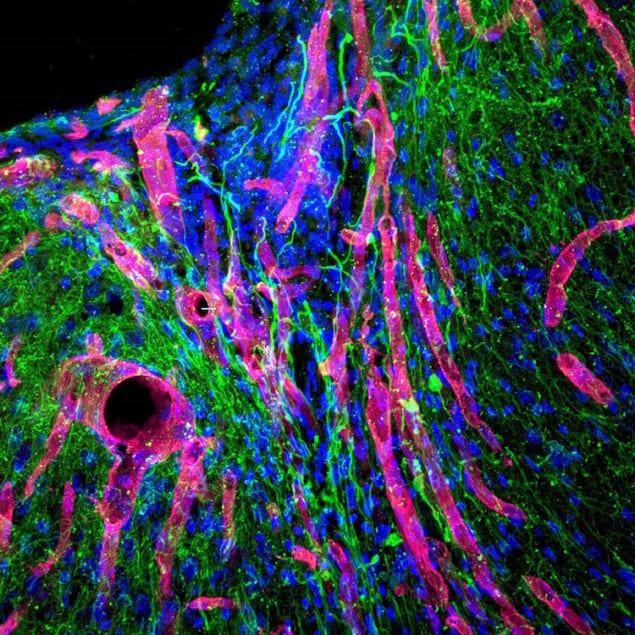
Stroke is one of the leading causes of death and disability worldwide and an estimated six million people in the US alone are living with its debilitating after-effects. The brain only has a limited capacity to regenerate following a cerebrovascular attack, as it is also known. This is because stroke damages brain tissue and produces a “dead” cavity devoid of blood vessels, neurons or axons (the thin nerve fibres that protrude from neurons). Researchers at the University of California, Los Angeles, have now developed a bioengineered angiogenic gel that promotes new brain tissue growth as well as the formation of axonal networks along regenerated blood vessels when it is injected directly into the stroke cavity of mice. The gel contains molecules that stimulate the growth of these blood vessels, explain team leaders Tatiana Segura and S. Thomas Carmichael.
The UCLA researchers made their biomaterial from an amorphous non-fibrous hydrogel composed of hyaluronic acid, the vascular endothelial growth factor (VEGF) and heparin nanoparticles (to bind the growth factor molecules). Once injected, the gel thickens to mimic the properties of brain tissue so creating a “scaffold” for tissue growth.
Promoting tissue repair following stroke
“This Clustered VEGF Nanoparticle (CLUVENA) Hydrogel, as it is called, promotes tissue repair following stroke through at least two mechanisms,” says Carmichael. “It induces angiogenesis and subsequent in-growth of neuronal connections in the stroke cavity. The heparin nanoparticles also reduce inflammation and scarring around the damaged stroke site and enhance the vascular and neuronal growth processes.”
“Both features of the material are required and necessary for brain repair to occur,” adds Segura, who is now at Duke University.
In a normally developing body, blood vessels and nerves grow together into tissues as they mature, grow larger and begin their adult function, continues Carmichael. There is a complex signalling system between nerves and blood vessels in this process. By stimulating angiogenesis and then maturation of new blood vessels, local brain cells are “attracted” to these vessels and grow connections along them. This is known as a neurovascular interaction.
Potential new therapy for stroke
“Our work importantly shows that the nanoparticle clusters containing VEGF induce a specific and very different pattern of VEGF signalling than that seen when VEGF is simply injected by itself into a stroke cavity,” he tells Physics World. “This clustered VGEF promotes organized and mature vessel networks that are likely key in secondary processes of nerve in-growth,” adds Segura.
The study, which is detailed in Nature Materials, also shows that post-stroke tissue is not irreversibly degenerated, she says. “A biomaterial like the one we have developed could thus be injected into the stroke cavity following an attack to regenerate brain tissue and promote recovery. This could make for a potential new post-stroke therapy.”
In the experiments on mice, the researchers found that stroke cavities in the animals contained regenerated brain tissue after 16 weeks, including new neuronal networks. This has never been seen before, they say. “The mice with new neurons also showed improved motor behaviour, but we are unsure of the mechanism behind this,” say Carmichael and Segura.
“The new axons could actually be working, or the new tissue could be improving the performance of surrounding, unharmed brain tissue.”
This study focused on the period immediately following chronic stroke (five days in mice, which translates to two months in humans). The UCLA team will now be looking into whether the biomaterial works as well after longer delays.



