Researchers in Switzerland and Israel have succeeded in fabricating the most efficient metal-oxide photoanode to date for use in photoelectrochemical cells (PECs) – devices that produce hydrogen using sunlight in a process called “water splitting”. The novel feat, which involved accurately characterizing the iron-oxide nanostructures used in PECs, could help in the development of much more efficient solar cells in the future.
Solar water splitting, in which water is separated into oxygen and hydrogen using sunlight, could be a clean and renewable way to produce energy. Researchers are busy looking for efficient photoelectrode materials for use in this process. One such material is haematite (rust). Iron is cheap and abundant, and haematite has a high theoretical solar-to-hydrogen conversion efficiency of 14–17%. However, it is still unclear how defects in this material affect its ability to convert solar energy into hydrogen fuel and why some haematite electrodes appear to be more efficient than others.
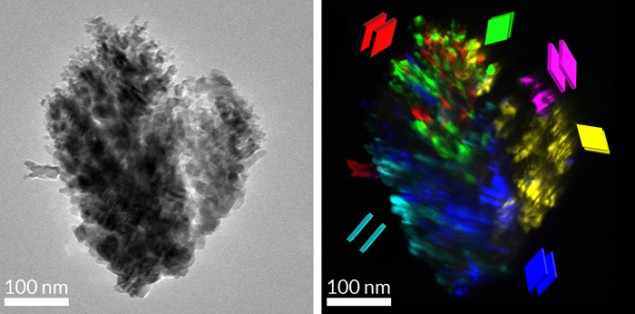
In an effort to answer these questions, a team led by Michael Graetzel of the Ecole Polytechnique Fédérale de Lausanne and Avner Rothschild of the Israel Institute of Technology decided to investigate how structure is related to performance at the single-nanostructure level in haematite. In an individual, centimetre-sized water-splitting electrode there can be billions of individual nanostructures, explains team member Scott Warren. Until now, however, researchers have typically studied the structure and properties of these nanostructures in aggregate and neglected individual structures. “We have shown that there are important differences among the individual nanostructures in a single electrode – differences that determine whether a nanostructure is active for water splitting or entirely inactive,” says Warren.
New performance record for water splitting
By understanding these differences, the team then developed synthesis techniques that allowed it to make the most efficient haematite nanostructures for water splitting. The researchers used these “champion” nanostructures to fabricate photoelectrodes capable of generating a photocurrent of about 4 mA cm–2. This is the highest photocurrent ever achieved for haematite and, indeed, any metal-oxide photoanode, as well as a new performance record for water splitting, says Warren.
“Our approach involves looking at a single nanostructure and determining how its structure is unique,” explains Warren. “We then measure how current moves through that single nanostructure and sometimes find that no current passes through. This is because of structural defects that we identified in a transmission-electron-microscopy technique (TEM) developed in our lab [see images]. We can then begin to understand what aspects of a structure impact on current transport. It is this sort of information that was inaccessible to researchers in the past.”
In analogy to the best performing champion solar cells, the Swiss–Israeli team has shown that some nanostructures are great at water splitting and others less so. The distinguishing characteristic of the nanostructures identified in this study is that all of the haematite crystals within the nanostructure are orientated in the same direction – something that allows electrons to travel rapidly through the material.
The research could help make better batteries, solar and fuel cells, claims Warren, although there is still much work to be done. “While the haematite we looked at performs better than any other equivalent cheap and stable material, we still need to improve its performance,” he says. “Continuing to characterize the nanostructure of haematite in this way will help us identify defects and other bottlenecks hampering its solar water-splitting efficiency.”
The research is published in Nature Materials.



