
In 2014, over 422 million people worldwide were suffering from diabetes mellitus. Three types of diabetes exist, and all affect the production of insulin, either because the pancreas cannot produce enough insulin, or due to an inappropriate response of the cells to insulin, a hormone that is secreted in response to high glucose in the blood.
Diabetes, due to high levels of glucose in the blood, can cause neurologic, ophthalmologic, nephropathic and cardiologic disorders, as well as birth defects in infants born from diabetic mothers. Diabetic patients need to assess their glycaemia several times per day, using a blood glucose self-testing device. Despite the existence of non-invasive devices, this is challenging for the patient. An alternative to blood glucose monitoring would be of great interest for diabetic patients.
With this aim, Joseph Lakowicz and his team at the University of Maryland have developed a more convenient way to read glucose, from tears, using glucose-sensitive silicone hydrogel contact lenses. Glucose concentration in tears follows the concentration in blood within five minutes, so such measurements should be accurate (J. Biomed. Opt. 23 057005).
The presence of an interface between the silicone and water regions in a typical silicone hydrogel lens provides the opportunity to create the glucose-sensitive lenses. The water regions act as channels for tear fluid transport and the silicone-rich regions allow a high transport of oxygen, since the capacity of the lens to transport oxygen defines the duration of its wearability by the patient.
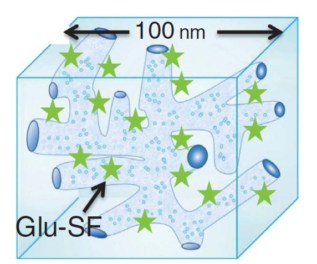
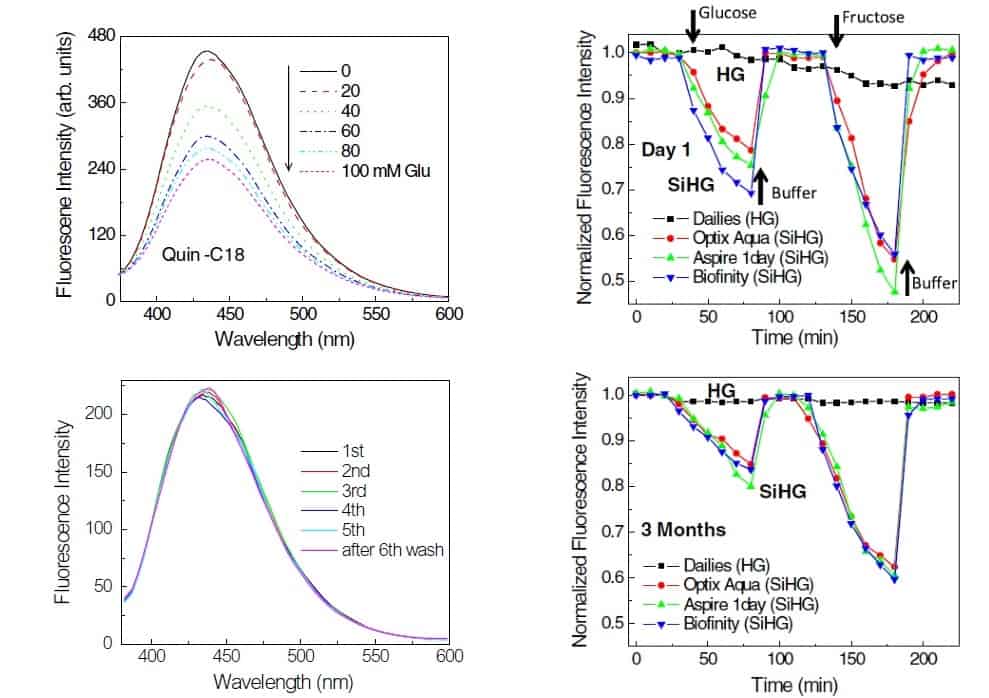
The system works thanks to the glucose-sensitive fluorophore Quin-C18, which contains a boronic acid that binds sugars. Quin-C18 fluorescence can be observed under an ultraviolet lamp. However, the device that will be used to detect the fluorophore in the lens is not yet determined. New technology such as a complementary metal–oxide–semiconductor (CMOS) camera could be used.
Lakowicz, and co-authors Ramachandram Badugu and Edward Albert Reece, observed that the fluorescence of the fluorophore Quin-C18 decreased when the concentration of glucose increased. There was no signal loss from the fluorophore after multiple rinses. Also, long storage (three months) did not seem to affect the lens’ response to glucose.
The researchers also demonstrated that glucose binding is reversible, by looking at the binding of fructose, which has a high affinity for boronic acid, after glucose binding. However, the minor amount of fructose in tears is not expected to affect the glucose measurements.
Besides their important potential for monitoring glucose in tears of diabetic patients, these lenses could also be used to detect other molecules or pathologies of the eye, for example for diagnosis of dry eye disease.



