Photon-counting CT employs direct conversion detectors to deliver high‑resolution, low‑dose spectral imaging. Sophie O’Neill takes a look at the potential of this emerging imaging technology.
Photon-counting computed tomography (PCCT) is an advanced medical imaging technique that differs from conventional X-ray CT in that it can discriminate between the energies of individual detected photons. Offering higher spatial, spectral and contrast resolution than conventional CT, PCCT could deliver significant benefits for disease characterization and enable new diagnostic approaches.
Conventional CT measures the attenuation of X-rays after they pass through the body, enabling clinicians to monitor normal and abnormal anatomy and providing valuable information for diagnosis and treatment of disease. The advantages promised by PCCT primarily arise from the differing characteristics of the detectors: conventional CT scanners use energy-integrating detectors (EIDs) whilst PCCTs employ photon-counting semiconductor detectors.
The effective dose from diagnostic CT procedures is estimated to be in the range of 1–10 mSv, although this can vary by a factor of 10 or more depending on patient size, the type of CT scan performed, the CT system and the operating technique. PCCT systems offer better dose efficiency than conventional CT and use energy thresholding to eliminate background electrical noise. As a result, PCCT requires lower radiation dose than standard CT – reducing the risk to the person being scanned.
Detector characteristics: limitations and advantages
Conventional CT systems use an EID to collect the total energy deposited by all incident X-ray photons. EIDs are typically composed of gadolinium oxysulfide (Gd2O2S) or cadmium tungstate (CdWO4) and comprise two layers: a solid-state scintillator placed on top of a photodiode array. The detection mechanism is a two-step, indirect process. Incoming photons hit the scintillation layer, which produces a flash of visible light. When the photodiode absorbs this light, it converts it into an electrical signal.
The photodiode array consists of individual detector elements separated by opaque, reflective walls called septa. This design prevents optical cross talk (signals transferring between adjacent channels and reducing image quality) produced by light scattering. The need for septa, however, creates “dead space” on the detector surface, which wastes X-ray dose and limits the spatial resolution since it physically restricts detector size.
As EIDs collect the total energy from all incoming photons, signals from photons of different energies are mixed together. High-energy photons will generate a higher light intensity than low-energy photons and will consequently produce a higher intensity electrical signal. This means that the final output signal will be dominated by the high-energy photons and under-weight the valuable contrast information that the low-energy photons provide. It also prevents the distinction between electrical noise and genuine low-energy photons, which further affects the achievable contrast.
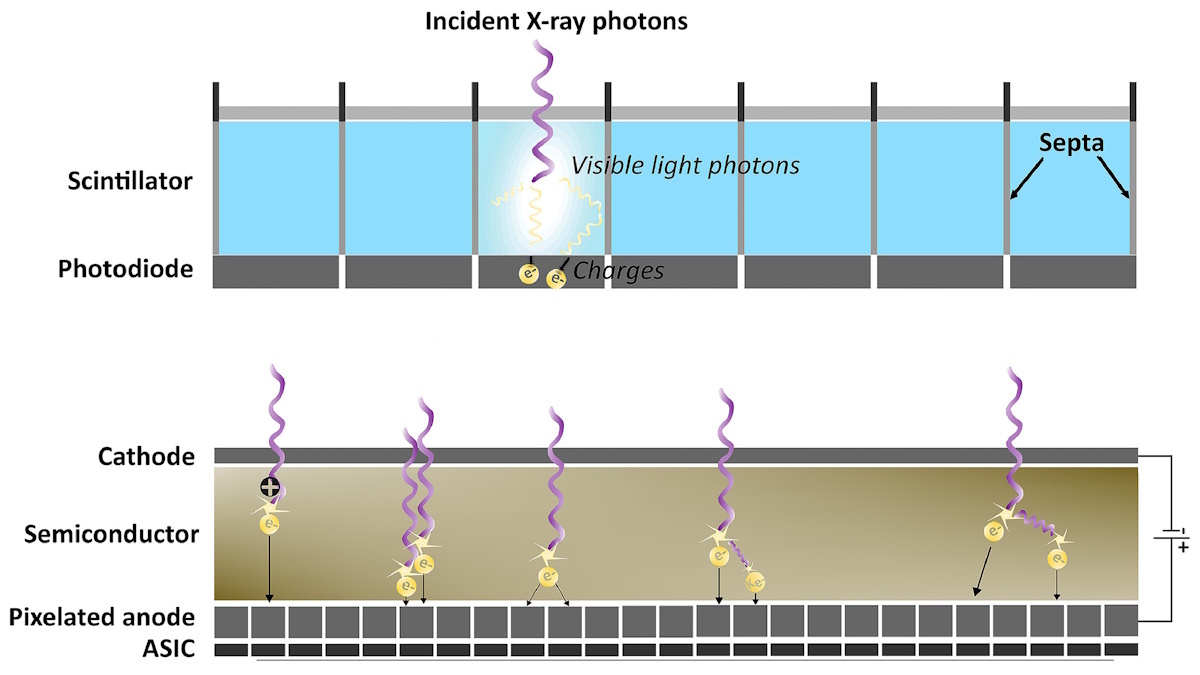
PCCT scanners, on the other hand, employ photon-counting detectors that directly convert the photon energy to electric signals. These detectors consist of a semiconductor layer placed between a cathode on the upper side and an anode underneath. The anodes are pixellated to increase spatial resolution, with each pixel placed on top of an ASIC.
This detector uses a direct conversion process in which a high bias voltage is applied across the semiconductor to generate electron–hole pairs when struck by an incoming photon. The strong electric field draws the clouds of charge toward the anode electrodes, creating a current. The ASIC instantly processes this current and converts it into a voltage pulse, with the height of the pulse directly proportional to the incident photon’s energy. Comparators and counters sort the photons into energy bins based on threshold values, a process that can also filter out electronic noise and enable spectral imaging.
The semiconducting materials used in photon-counting detectors are typically either cadmium telluride (CdTe), cadmium zinc telluride (CZT) or silicon. The cadmium-based detectors have high stopping powers due to their high atomic number, leading to efficient absorption of X-rays via the photoelectric effect and resulting in a high spatial resolution. Another advantage of CZT and CdTe detectors is that the semiconductor can be relatively thin (roughly 2 mm), allowing the detector to be placed perpendicular to the direction of the incident X-rays.
Advanced spectral capabilities
Conventional CT relies on post-processing software to enhance image resolution and reduce the electronic noise that’s inherent to its physical hardware. But the algorithms traditionally used for image reconstruction – which include back projection, filtered back projection and iterative reconstruction algorithms – can reduce spatial resolution and cause blurring.
Deep learning-based reconstruction, meanwhile, can induce artefacts (such as generating objects that don’t exist or removing true small anatomical structures), particularly in low-dose scenarios where training data are limited. To achieve high resolution in conventional CT, a low-energy filter in the X-ray beam is needed, which increases the required radiation dose.
The PCCT detector design, with small pixel sizes and lack of reflective septa, make it an inherently high-resolution technique. Image quality can be further improved using algorithms such as quantum iterative reconstruction, which has been shown to reduce image noise by up to 34.5%. Sharp convolution kernels (used to optimize the balance between noise and sharpness) are needed to ensure that the image produced maintains the high resolution provided by the detector.

The ability of PCCT to distinguish photon energy also allows for material decomposition, which enables the generation of a range of advanced images. This includes virtual monoenergetic images reconstructed at a single energy level to amplify contrast agents without reducing dose, and virtual non-contrast images, which allow digital subtraction of particular materials without needing another scan. PCCT can also be used for K-edge imaging, in which contrast agents can be isolated based on their isolation of their K-edge energies.
Clinical applications
The technical advantages of PCCT have significantly improved the diagnostic applications of CT across a plethora of medical disciplines.
For instance, a prospective study on 200 adults with lung cancer who underwent both PCCT and EID CT showed that PCCT outperformed conventional CT in lung cancer management. The key findings were that PCCT had a lower effective radiation dose (1.36 mSv) compared with EID (4.04 mSv), lower exposure to iodine (a dye used to increase image contrast), with an iodine load of 20.6 mSv for PCCT (compared with 28.1 mSv for EID CT) and higher detection and diagnostic confidence for enhancement-related malignant features.
Similarly, in a study of CT pulmonary angiography, PCCT reduced the total iodine load by 26.7% and the CT dose index volume by 24.4% compared with EID CT. This potentially lowers patient risk, as well as providing environmental and financial benefits.
Within coronary imaging, PCCT enables characterization of coronary artery disease and plaque and shows promise in coronary artery calcium quantification by reducing blooming artefacts (where small, high-density structures like calcium appear larger than their true size). PCCT can also provide high-resolution imaging of the lumen for evaluation of coronary stents and assessment of myocardial tissue and perfusion.
The higher dose efficiency of PCCT makes it particularly effective in paediatric applications, as children are more radiosensitive than adults. Children also have smaller organs, making the ultrahigh resolution provided by PCCT especially helpful, for example, in the detection of tiny, complex heart defects in neonates and infants.
As of early 2025, there were two US Food and Drug Administration (FDA)-cleared PCCT systems in clinical use: the NAEOTOM Alpha from Siemens Healthineers and Samsung Healthcare’s OmniTom Elite. And just last month, the Extremity Scanner System from MARS Bioimaging and GE HealthCare’s Photonova Spectra photon-counting CT both received FDA clearance. Other clinical prototypes include systems from Canon Medical Systems and Philips Healthcare.
Ongoing challenges
As with any emerging technology, challenges remain to be solved. With photon-counting detectors, these includes effects such as pulse pile-up, charge sharing, K-escape and Compton scattering.
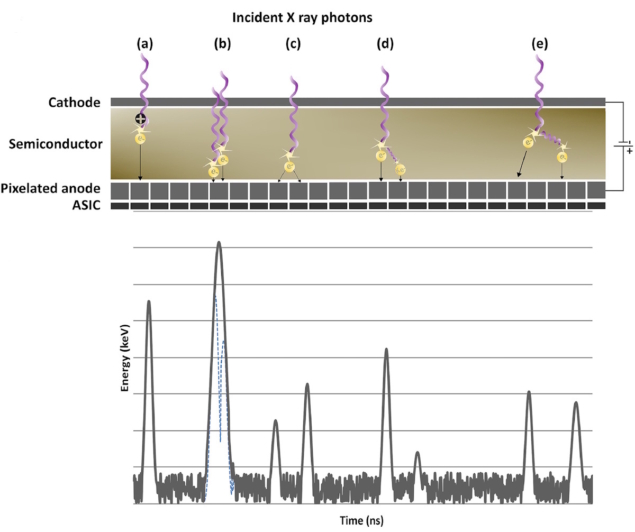
Pulse pile-up occurs when two or more photons arrive at the detector simultaneously, which may result in it recording this as a single photon. This leads to errors in the calculation of energy received at the detector and determination of the numbers of photons. If a single photon strikes near the boundary between two pixels it may be detected as having lower energy than it actually has. This effect, known as charge sharing, will degrade the spectral and spatial resolution of the CT image.
Due to their high atomic number, cadmium detectors are also susceptible to an effect known as “K-escape”, in which incident X-rays produce fluorescence that’s detected as a separate event. Compton scattering occurs when a secondary photon produced in the semiconductor material is registered as a separate event, underestimating the real energy value.
Finally, manufacturing the semiconductor materials used in PCCT is expensive – PCCT scanners can cost in excess of £2 million. And the large data sets generated by multi-energy scanning require a large amount of computing power and time to process and reconstruct.
Future impact
PCCT is a highly promising technology that replaces traditional indirect detection mechanisms with direct detection using semiconducting materials. PCCT offers superior image quality due to higher spatial and spectral resolution, higher dose efficiency and the ability to perform quantitative imaging. The multi-energy capabilities of PCCT shift the image from providing purely structural information to also include functional information.
Current clinical use is limited mainly due to cost rather than diagnostic capability, with a lack of clinical studies making the high cost difficult to justify. However, the potential impacts for optimizing healthcare could be vast. Perhaps it is inevitable that, as costs decrease with evolving technology, the clinical use of PCCT will overtake conventional CT in the future and become the standard CT technique.