Bringing together clinicians, basic scientists and industry experts can be a challenge, but it’s crucial for translating scientific advances into clinical benefits. Steve Smye explains how the National Institute for Health Research supports this process
The UK is a world leader in life-sciences research. The life-sciences industry is also increasingly regarded as a vital part of the nation’s economy, with the government’s Industrial Strategy (published in November 2017) setting out many ambitious goals for the sector. However, until recently the process for translating top-class research and innovations into benefits for patients in the National Health Service (NHS) was not well developed. While the NHS has a long history of conducting research funded by research councils and medical charities, its track record of effective and appropriate working with industry partners has historically been much patchier. As little as a decade ago, both commercial and non-commercial research partners regularly complained of lengthy delays in setting up studies, and a relatively high proportion of studies failed to recruit enough patients to arrive at robust conclusions. It was clear that a much more systematic approach to conducting clinical research was needed.
This was the situation in 2006, when my organization, the National Institute for Health Research (NIHR), was founded with a mission to “improve the health and wealth of the nation through research”. It does this by funding high-quality research; training and supporting health researchers; providing world-class research facilities; and working with the life-sciences industry and charities across England. Crucially, our work involves patients and the public at every step: more than 650,000 patients participated in NIHR-recognized research studies in 2016/17. In terms of industry support, we give life-science firms unparalleled access to (and understanding of) the NHS research environment, helping them with study feasibility analysis, set-up, costings, contract negotiation and performance monitoring. This support covers a wide range of research types, from early-stage, translational research through to later-stage clinical trials in the NHS. By working collaboratively with the life-sciences industry in this way, the NIHR helps patients gain earlier access to breakthrough treatments and encourages broader investment in (and economic growth from) health research.
Improving radiotherapy
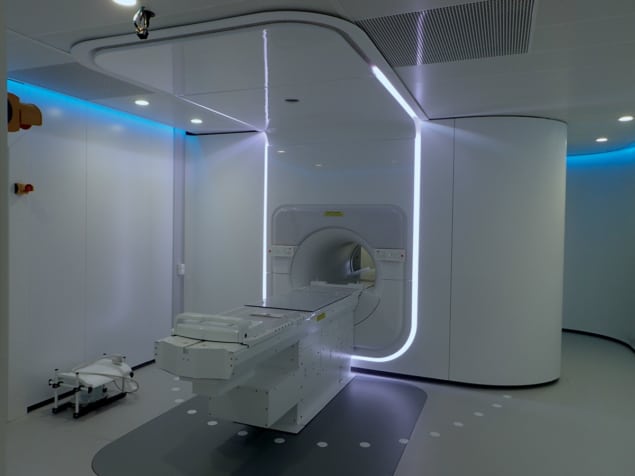
Although the initial focus within the NIHR was, understandably, on drug trials, we are now working with commercial partners from the pharmaceutical, biotechnology, diagnostics and medical-technology industries, as well as contract research organizations. As an example, consider the NIHR Manchester Biomedical Research Centre, which is part of a network of organizations set up to conduct experiments in areas that include medical imaging and radiotherapy. One of the Manchester centre’s projects is to identify and develop biomarkers that can predict the effectiveness of different types of radiotherapy and drug-radiotherapy combinations, while minimizing the risk of long-term side effects. The centre’s researchers are also working with industry and outside experts to overcome the operational challenges (such as build, installation, calibration and use) associated with embedding new technology in a clinical setting. In particular, they are part of an international consortium supported by Elekta, a major manufacturer of radiotherapy machines that is developing guidelines for targeting tumours more accurately using linear accelerators equipped with magnetic resonance imaging (MR-linacs).
Current radiotherapies are already personalized and adaptive to some degree, with treatments based on the size and shape of both the individual and the tumour, along with the tumour’s location. Depending on the latter, patients may be scanned during their radiotherapy and their treatment may be adjusted accordingly. MR-linacs have the potential to take this personalization a step further, because they make it possible to image the patient at the same time as each dose or “fraction” of radiotherapy is delivered (“see while you treat”). This means that clinicians can create adaptive radiotherapy plans that are fine-tuned to daily changes in the patient’s anatomy – something that could revolutionize cancer treatment by lowering the radiotherapy dose to surrounding organs, thereby reducing side effects and improving patients’ quality of life.
There is, however, one drawback, which is that the strong magnetic field of the MR scanner affects radiation treatment. Researchers on the MR-linac team at Manchester have recently published a review article (Clin. Oncol. 29 662) describing the benefits and challenges of introducing this technology, and setting out progress to date. The review highlights how the MR-linac’s superior imaging capabilities when compared to current technologies (notably cone-beam CT imaging) will enable treatment plans to be adapted while a course of radiotherapy is being delivered. It also discusses the difficulties of developing imaging protocols for certain areas of the body, such as the lung, that are harder to image with MRI machines.
Tests and technologies
Both radiotherapy and imaging are traditionally very strong areas for physics-led medical research. Increasingly, however, they are not the only fields where physicists and engineers are playing prominent roles (often in partnership with industry) in the NIHR’s work. In September 2017, for example, the NIHR began setting up 11 new centres – known as medical technology and in vitro diagnostics co-operatives, or MICs – dedicated to developing technologies and tests related to conditions such as kidney and liver disease where patient morbidity is high. One of these MICs is at the Leeds Teaching Hospitals and University of Leeds. Its scientific director, Steve Evans, specializes in molecular and nanoscale physics and is developing physics-based tools for characterizing single cells. The position of the Leeds MIC at the interface between the physical sciences and medicine is exemplified by its work on novel nanomedicines for cancer therapy (see image below). The involvement of numerous industry partners in the Leeds MIC is typical of the programme as a whole.

Another area where physicists are getting involved in health research is the NIHR’s Invention for Innovation (i4i) programme, which funds translational research into healthcare technologies, devices and interventions that could benefit patients in areas of existing or emerging clinical need. The programme aims to reduce the risk of embarking on such projects, thereby making them more attractive to follow-on funders and investors, and the expected outputs are advanced or clinically validated prototypes.
Nick Stone, a medical physicist at the University of Exeter who has used this “pot” of funding for several projects, calls it “the ideal funding stream to enable us to translate our novel technologies into real clinical tools”. Stone’s most recent i4i-funded research project has been carried out in collaboration with experts at the University of Bristol and Gloucestershire Hospitals NHS Foundation Trust, and it uses lasers to detect oesophageal cancer. This type of cancer is often discovered so late that treatments (even successful ones) are very distressing, dangerous and difficult, but Stone and his team have found a way to tell the difference between healthy and diseased tissue by shining a low-power laser on the tissue and looking at the resulting inelastically scattered light – a technique known as Raman spectroscopy.
Before receiving the i4i funding, Stone and his collaborators had designed a miniature probe that slides through a channel in an endoscope and onto the surface of the oesophagus. In the lab, this device can assess the condition of oesophageal tissue almost instantly, determining whether it is healthy, pre-cancerous or cancerous without the need for biopsies. The i4i project currently underway aims to finish developing the probe and begin assessing the effectiveness of Stone’s technique in actual patients. The hope is that if this work can be translated from the lab into clinical practice, doctors will have a new way of diagnosing oesophageal cancer and pre-cancerous lesions much earlier, when treatments are more likely to be effective.
Physicists as brokers
I am a physicist myself, and although medical physicists and engineers comprise a relatively small proportion of the NHS workforce, I am passionate about promoting the value of a “physics-based” approach to medicine, in which quantitative models of disease are increasingly prominent. In the NHS, physicists can also play an important role as “brokers” between medical staff, basic scientists and industry experts. Another exciting development is the “Physics of Life” network, which is jointly funded by the Engineering and Physical Sciences Research Council and the Biotechnology and Biomedical Sciences Research Council, and which shows promise in promoting research at the interface between the biological and physical sciences. The Institute of Physics’ Biological and Medical Physics groups also have well-established programmes of multidisciplinary activity.
For such networks to function effectively, however, we need scientists who are enthusiastic about working across boundaries between disciplines and sectors; willing to spending time understanding the underlying science; and open to applying approaches developed for one discipline to other areas (an excellent and contemporary example of this is the application of statistical physics to problems in biology). Interdisciplinary areas such as medical physics and engineering, biological physics and biomathematics have come of age in recent years, and if we are to truly exploit their scientific potential, we need to continue the drive to create vigorous interdisciplinary networks.
Although the rewards of working with good medical collaborators are immense, collaborating with – or even finding! – busy clinicians is often fraught with frustration and difficulty. The demands on medics’ time are significant, so would-be collaborators – whether they are academics doing basic research or industry partners seeking clinical advice – need to adopt some simple, practical steps to ensure that collaborative discussions are made as easy as possible. In many circumstances, the best approach for UK-based collaborators will be to link to one of the elements of the NIHR infrastructure (see www.nihr.ac.uk), or its equivalent in Wales, Scotland or Northern Ireland. Academic physicists and industry partners who do this will find that there is plenty of enthusiasm to work together to realize the benefits of physics in medicine.



