Two particles carrying electrical charge with the same sign should not attract each other, but in recent years, researchers have found that they can do this when they are dispersed in a liquid. A team at the University of Oxford in the UK has now discovered that the distance over which this counterintuitive “electrosolvation” force acts is much longer than theoretical models currently predict. They have also shown that the range of the force can depend on particle properties such as size and surface chemistry.
“The new finding reveals a missing piece in our understanding of electrostatic forces in liquids,” says physical chemist Madhavi Krishnan, who led this research study. “It is likely to reshape our understanding of how biological matter may self-organize and how molecules like DNA, RNA and proteins may naturally condense and cluster inside cells.”
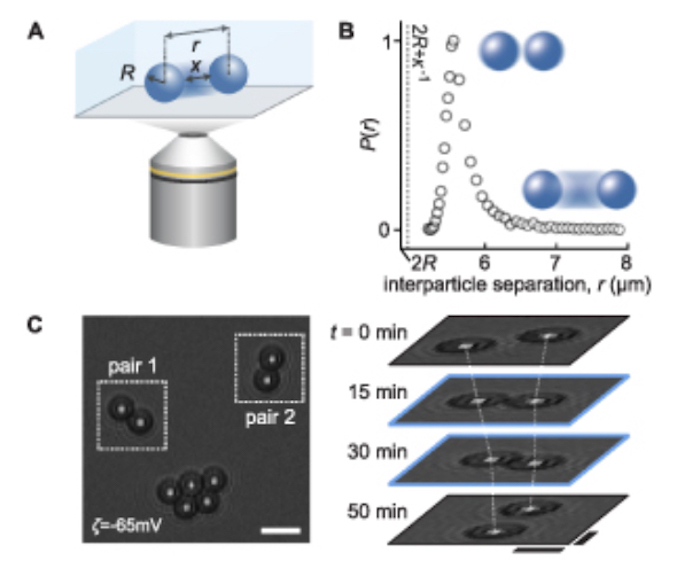
In their work, Krishnan and colleagues used optical imaging to observe how pairs of charged micron-sized spheres with various surface coatings, such as DNA, polypeptides and anionic lipid bilayers (which make up cell membranes) interact in water.
Not a uniform medium
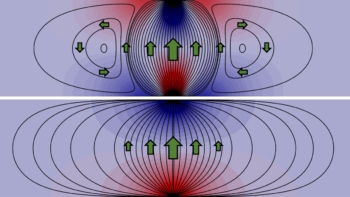
“Conventional electrostatic models treat the solvent as a uniform medium with a dielectric constant, but real liquids (such as water) cannot be described in this way because they form hydrogen bond networks and orient themselves around surfaces. Liquids also exhibit long-range correlations. All these properties may play a role in giving rise to an additional force which we call the electrosolvation force,” explains Krishnan.”
To be able to come up with a comprehensive understanding of the electrosolvation interaction, we have to dissect and carefully examine the phenomenology in question, she explains. A key feature of an interaction is its range. To measure the range of the attractive electrosolvation force accurately, Krishnan says that the students who carried out the experiments – her graduate student Sida Wang in particular – performed careful microscopy measurements on particles interacting with each other, observing individual pairs for periods of up to an hour and sometimes longer.
“We also performed exhaustive computer simulations to vet the measurements and estimate their accuracy,” Krishnan adds.
The researchers observed that DNA-coated particles exhibit particularly long-range attraction, which implies that the interaction depends not only on the solvent but also on the chemical and physical structure of the particles’ surface. This contrasts with the long-held view that the (Debye) screening length governing the interaction of charged particles in solutions depends only on the properties of the solvent medium.
Krishnan explains that the measured range of the attractive electrosolvation force can significantly exceed the nominal Debye length is to our knowledge not readily accounted for within any existing theoretical view and points to major gaps in our understanding of this very basic and fundamental question of how two charged particles interact in a liquid.. Indeed, it highlights the need for a more sophisticated view of the intervening medium than that offered by standard continuum electrostatics models.
“Current electrostatic models are incomplete”
“In short, anionic matter seems poised to attract; and the ability to either attract or repel in water, depending on the conditions, appears to be an intrinsic feature of negatively charged matter,” she tells Physics World. “It is entirely possible that the underlying mechanisms behind this process are broadly exploited in biology.”
Scientists discover that like-charged particles can sometimes attract
The new work, which is detailed in Reports on Progress in Physics is the most recent in a series of investigations on the physics of interparticle interactions in the fluid phase, she says, and once again shows that current electrostatic models are incomplete – even under conditions in which they are expected to work well.
Looking ahead, the researchers say they would now like to examine the same interactions in bulk solution and compare these observations in the sedimented colloids studied in the present work.