
Researchers from South Korea and Switzerland have developed magnetically actuated microrobots and used them for precise stem cell delivery and transplantation in vitro, ex vivo and in vivo. They also demonstrated that the microrobots could transport cancer cells through a body-on-a-chip model of a liver tumour network (Sci. Robot. 10.1126/scirobotics.aav4317).
Stem cell therapy is an emerging method for restoring damaged tissues or treating a variety of diseases, from cancer to degenerative neural disorders like Alzheimer’s disease. Precise delivery of stem cells to the desired target is, however, crucial for the success of transplantation. Small, minimally invasive and wirelessly controlled, magnetic microrobots have ideal qualities for use in stem cell therapy, but must be further improved before application in the clinic, the authors say.
In this work, Sungwoong Jeon from the DGIST-ETH Microrobotics Research Center and colleagues combined various pre-existing strategies of biocompatible microrobotic design to create spherical and helical microrobots. Fabricated by 3D laser lithography, the microrobots’ porous structure facilitates the attachment, proliferation and differentiation of various stem cells.
Upon application of a rotating magnetic field, the spherical and helical microrobots respectively exhibited rolling and corkscrew motions that propelled them through 3D spaces — such as a blood vessel isolated from a rat brain and a ventricle within a slice of mouse brain — enabling precise targeting of transplanted stem cells. The authors note that propulsion via rolling or corkscrewing motion is more efficient than that generated by a pulling motion and, as such, is more suitable for use in biological fluids.
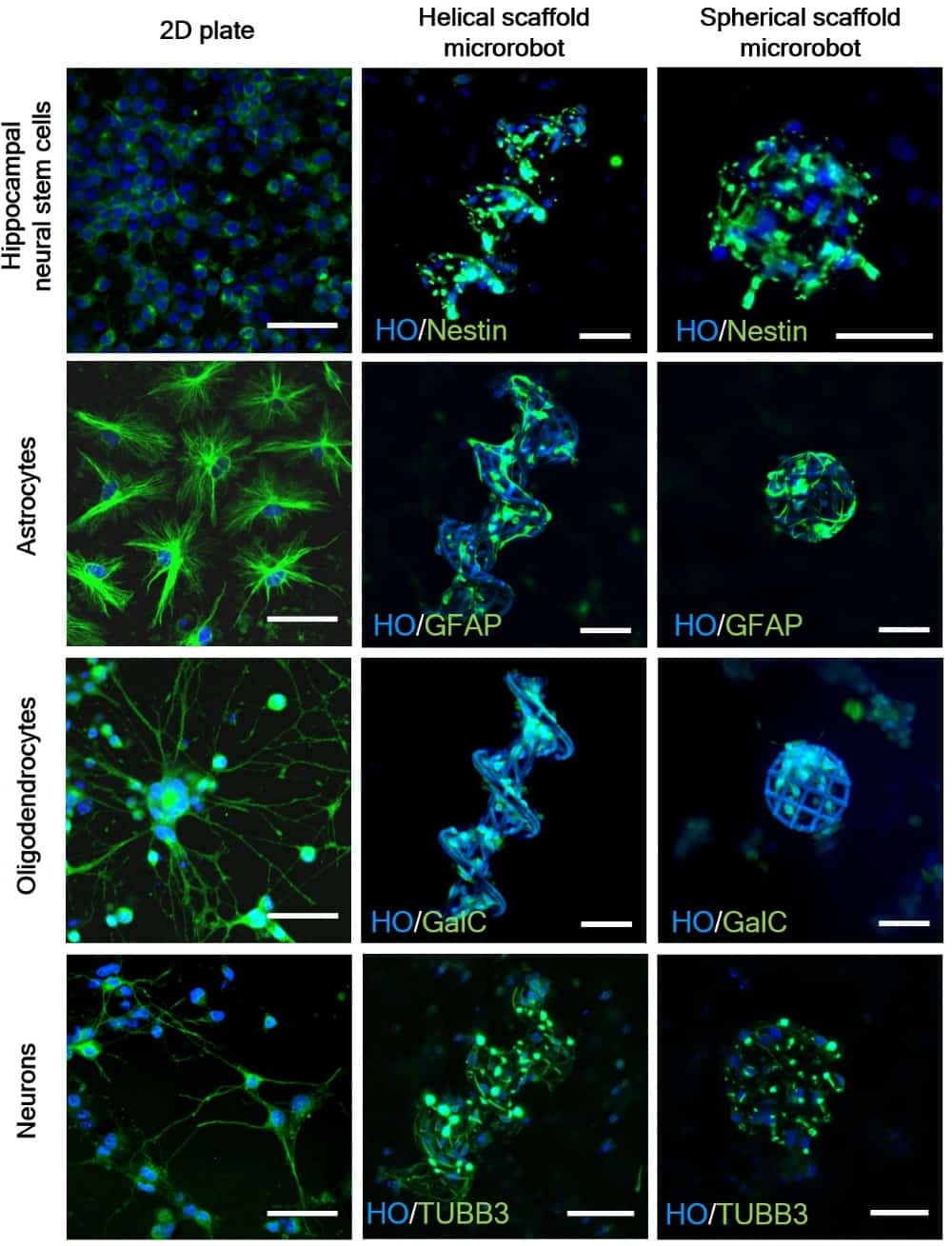
The researchers examined hippocampal neural stem cells (NSCs) grown on the microrobots. Immunofluorescence staining showed that, after 72 hours, NSCs on the microrobots differentiated into astrocytes, oligodendrocytes and neurons. Scanning electron microscopy revealed that the differentiated neurons became attached to and entangled with the microrobot scaffolds. The team also demonstrated magnetic manipulation of helical and spherical microrobots with attached hippocampal NSCs.
Next, Jeon and colleagues established the feasibility of targeted cell delivery using microrobots loaded with human colon cancer cells in a body-on-a-chip microfluidic cell culture platform that mimics parts of a liver tumour network. Applying an external magnetic field enabled the microrobots to be moved to a target location in the body-on-a-chip.
Finally, the team demonstrated in vivo stem cell transportation using the microrobots in a live nude mouse. They cultured mesenchymal stem cells derived from the human nose on spherical microrobots for transplantation in the mouse, and showed that an externally applied magnetic field could manipulate the cell-loaded microrobots within the animal’s hollow abdominal cavity.
These results confirm the feasibility of using microrobots for targeted stem cell transportation and transplantation in various physiological fluidic environments. In future work, the researchers hope to improve the controllability of the microrobots and reduce the strength of the magnetic field required to manoeuvre them.



