Neuro-radiosurgery is evolving to become more effective and less destructive to healthy brain tissue. One emerging approach is X-ray microbeam radiotherapy (MRT), a spatially fractionated radiation delivery method being investigated for the treatment of malignant brain tumours. MRT irradiates tissue with a synchrotron beam reshaped into arrays of highly collimated, micrometre-thick microbeams. The approach administers spatially-restricted peak doses of up to hundreds of gray to both healthy and tumour tissues
To study the effects of MRT, a European research team has used X-ray phase contrast CT (PCI-CT) – an experimental high-resolution imaging technique – to perform ex vivo studies of normal and cancerous rat brain morphology after treatment at the European Synchrotron.
The researchers found that PCI-CT could detect the effects of MRT throughout target tissue areas and distinguish cancerous tissue morphology, necrosis, and intra-tumour accumulation of calcium and iron deposits. It visualized brain anatomy and micro-vasculature in 3D. Importantly, PCI-CT displayed high soft-tissue contrast without requiring a contrast agent, enabling effective 3D segmentation and renderings of complex brain structure (Int. J. Radiat. Oncol. Biol. Phys. 10.10.1016/j.jirobp.2018.03.063).
High-resolution insight
For the study, Giacomo Barbone from Ludwig Maximilians-Universität München and co-authors implanted glioblastoma cells in the brains of six rats. They treated these rodents plus three healthy controls with a variety of different MRT protocols.
Following MRT, the rats were sacrificed 45 days later, and their brains were extracted for imaging. The researchers then acquired PCI-CT image datasets with 3000 projections over 360° in half acquisition, giving an overall horizontal field-of-view of 30 mm. They reconstructed images using standard filtered back-projection algorithms and computed brain vessel 2D maximum intensity projection (MIP) maps. To obtain a comparable MRI dataset to validate PCI, the authors imaged one cancerous brain sample with a pre-clinical 9.4T MR scanner.
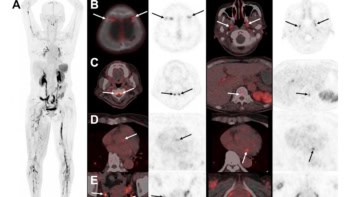
The authors reported that the full structure of a healthy brain (including cerebellar, cortical, thalamic, and hypothalamic structures) was well visualized in both sagittal and coronal slices. Brain sub-region anatomy could be visualized in the coronal slices.
They observed cancerous brain tissue within striatum, thalamic and hypothalamic regions, as well as inside lateral vessels. These were visible as regions of higher phase-contrast relative to normal brain parenchyma. The PCI signal within glioblastoma tissue was generally higher than in normal nervous tissue. Necrotic areas in the centre of tumour masses generated low PCI signal. Calcium and iron accumulation within degenerating cancerous tissues created intense bright signals in PCI.
The researchers identified brain blood vessels in normal and tumour-bearing tissues throughout the PCI data. They were able to segment hyper-dense features to create 3D vessel-network trees, and create 2D MIP maps by superimposing several consecutive image slices. Larger superficial blood vessels in the subarachnoid space were visualized as bright tubes, and intra-cortical vasculature and capillaries as bright tubules. Deep hyper-dense microvascularization had the visual appearance of either an organized network or a “chaotic bundle” wrapping around the glioblastoma in a tumour-bearing cerebrum.
Tissue effects caused by MRT were also well visualized. Brain tissue was marked with 50 μm parallel lines of ablation from the spatially-fractionated microbeams, and hypo-dense comb-like patterns were seen. PCI clearly showed the morphology of MRT-targeted local vasculature and microvasculature, as well as intra-cortical microvasculature, cancerous lesions, MRT-driven tissue ablations and minute cerebral structures.
One potential benefit of PCI is that it does not require extensive sample preparation. The sample does not need dissection, making it well-suited for some post-mortem morphological brain analyses. In addition, PCI concurrently visualizes tumour tissues, healthy tissues, micrometric angio-structure, and the effects of high-dose ionizing radiation in a “one-shot” image.
The researchers believe that PCI could be employed to precede and guide histological analysis. Because PCI can quantify tissue volume modifications, the technique could potentially also be used for studies of brain tumour tissue radio-resistance or to assess drug efficacy in experimental models of neurodegeneration.