A technique that identifies malaria infections in blood using cheap magnets and run-of-the-mill pocket lasers has been developed by scientists in Hungary. It exploits the unique magnetic and optical properties of crystalline waste produced by malaria parasites in the bloodstream and offers an inexpensive, sensitive and reliable alternative to existing diagnostic tools.
Malaria is the world’s number-one vector-borne infectious disease. It is contracted by 200 million people each year and proves fatal for one million of those; yet it is easy to treat, making many of these deaths avoidable. To date, medical science has come up with sensitive, equipment-heavy diagnostic tests as well as cheaper portable tests with lower sensitivity and accuracy – but none meet all the needs to efficiently combat the disease.
It was a 2008 paper, by Dave Newman and colleagues at the University of Exeter in the UK, describing a way to exploit the magneto-optical behaviour of “haemozoin” – a crystalline substance excreted by malaria parasites – that first caught the attention of István Kézsmárki, of the Budapest University of Technology and Economics and the Hungarian Academy of Sciences. When the parasites digest haemoglobin, they are left with a substance known as “haem” that is highly toxic to them, until they convert it into insoluble haemozoin microcrystals – also known as malaria pigment.
Unique properties
“The crystals are quite unusual…when the parasites turn haem into malaria pigment, it becomes magnetic,” explains Kézsmárki. “There is no other material in human blood that would have the same properties and produce the same effects.”
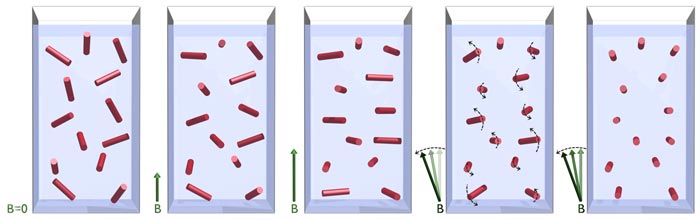
This is down, in part, to the dimensions of the crystals and how highly anisotropic they are at a molecular level. A crystal’s orientation governs the absorption or scattering intensity of incident polarized light on it.
So, placing an infected blood sample in a strong magnetic field forces all the crystals, which are normally thermally buffeted and jostled by surrounding molecules, to point in the same direction. Their collective effect on polarized light clearly points towards any malaria infection.
New twist
While these characteristics had already marked out haemozoin as ideal for use in malaria diagnosis, Kézsmárki and colleagues “made another twist to make it really feasible for cheap daily diagnosis”.
Instead of using research-grade instruments such as superconducting magnets and highly stable lasers, the researchers generated a uniform magnetic field by arranging a ring of standard €1 permanent magnets around the sample. By spinning the ring, they got the crystals to spin, with their moment of inertia coupled with the viscosity of the fluid, causing them to align in the field.
When the researchers shone a simple laser through the sample, the crystals acted as secondary polarizers, alternately transmitting and scattering light as they spun. A polarizing beamsplitter was used to separate the exiting light into its horizontal and vertical components. For uninfected blood, the intensities of the two components were the same, independent of the orientation of the magnetic field. For infected blood, the two oscillated inversely with one another as the magnetic field rotated.
Sensitive solution
Today’s best lab-based malaria test can identify parasite concentrations as low as 5/μL of blood, but it is too costly and impractical for large-scale diagnosis in rural areas where malaria is endemic. Rapid diagnostic tests, which involve no more than a drop of blood on a strip of antigen-coated plastic, are fast, portable, cheap and uncomplicated, but they have a sensitivity threshold of about 100 μL – too high to catch early-stage infection.
Kézsmárki’s team found that it could spot parasite concentrations as low as 25/μL of blood, and when the researchers ran the test on plasma instead of whole blood, their sensitivities jumped to unprecedented levels – one parasite/μL. Their method could potentially be applicable at the very earliest stage of the disease – the first symptom-free few weeks when the parasites have invaded the liver and are producing haemozoin but have not yet been dispatched into the bloodstream.
Also, haemozoin is extremely stable – the exact same form of the chemical is seen in fossilized remains of ancient malaria-infected creatures – and is common to all mutations of malaria. This means that the test will be viable in all locations and will never become obsolete, unlike the strain-specific rapid-detection tests, which face a constant struggle to keep up with the swiftly mutating malaria genome.
The real test
“The new method is exciting and may have the capability to be used as a rapid screening tool [once it is developed further],” says Stephen Karl of the Walter and Eliza Hall Institute in Australia, who was not involved in the research. He says the technique shows promise and anticipates that “the cost-per-test with this instrument will be very low since almost no disposable materials are required”.
David Bell, a malaria expert at the Foundation for Innovative New Diagnostics in Geneva, is a little more reserved. “It is difficult to tell at this stage,” he cautions, describing existing rapid-diagnostic tests as “adequate for case management”. To make an impact, he feels the new test will need to be “equally cheap, robust, without lots of moving parts, and not need batteries. That is a difficult ask”.
“The other question is whether you can determine one species from another by this method, which is important because different species of parasite need different treatments,” he adds.
For now, Kézsmárki and colleagues are looking to collaborate with engineers on reducing the size of the apparatus from its current “laptop” size to about 20 cm across, and are keenly pursuing the optimum way to separate red blood cells and plasma while keeping the malaria pigment intact in the plasma. “This is a crucial point to resolve,” Kézsmárki says. “Our target is to find the method that is the simplest, which requires no special biolab.”
The preprint of the research is available on arXiv.
- The upcoming November special issue of Physics World is devoted to “animal physics”. You can download a free PDF of the issue from physicsworld.com from Wednesday 7 November 2012
- David Hu from Georgia Institute of Technology’s laboratory for biolocomotion presents a special online lecture at 3.00 p.m. GMT on Thursday 8 November 2012, which you can view by registering here