Physicists in Sweden and France are the first to calculate the crystal structure of the metallic phase of silane — a normally gaseous compound of silicon and hydrogen that becomes a solid metal and then a superconductor when put under increasingly high pressure.
The result could be an important step towards understanding the structure of metallic hydrogen — an elusive material that is the “holy grail” of high-pressure physics because it could provide researchers with a new system to study high-temperature superconductivity.
A glance at the periodic table suggests that hydrogen should be a metal at sufficiently high densities because it is in the same column as the alkali metals — an idea that was proposed over 70 years ago by Eugene Wigner. Calculations done four years ago by Cornell University’s Neil Ashcroft also suggest that metallic hydrogen could be a superconductor at temperatures well above 30 K.
However, physicists haven’t had much luck in creating the metallic phase — solid hydrogen has been compressed to pressures as high as 320 GPa only to see it remain a semiconductor. This is about three million times atmospheric pressure and just about the limit of the diamond-anvil pressure cells used in such experiments. Scientists have had better luck with liquid hydrogen, which was shown to become a metal when compressed at 140 GPa by a shockwave — but this only lasted about 100 ns.
Hydrogen-rich materials
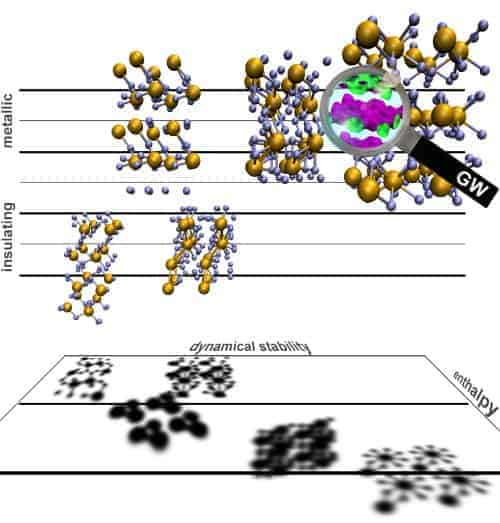
Instead of struggling to make metallic hydrogen, some physicists have turned their attention to the metallic phases of hydrogen-rich materials, such as methane (CH4) and silane (SiH4), which can be produced at much lower pressures. Such materials have electron densities similar to solid hydrogen and therefore could provide insights into the physics of metallic hydrogen.
Earlier this year, two independent teams of physicists compressed samples of silane and discovered that it became a metal at pressures greater than about 50 GPa — and became a superconductor when the pressure reached about 100 GPa. Although the positions of the silicon atoms in the metal was determined using X-ray diffraction, the precise crystal structure of the material could not be determined because it is almost impossible to “see” hydrogen atoms using X-rays.
Now, using state-of-the-art quantum mechanical calculations and a process of elimination, DuckYoung Kim of Uppsala University in Sweden and colleagues claim to have clearly identified the crystal structure of the metallic silane phase (PNAS 105 16454).
Theoretical ‘detective story’
The researchers have shown that a structure with space group P4/nbm should be metallic at pressures of more than about 60 GPa, which is in line with experimental results. Kim and colleagues’ work resembles a theoretical “detective story”: the scientists investigated a pool of plausible candidate structures and ruled out each structure that did not fulfil certain criteria that the true structure of metallic silane must possess. “This approach left us with a single structure by way of elimination, which we concluded to be the correct crystal structure,” explained Kim.
“Understanding metallic hydrogen-rich materials, such as silane, represents a stepping stone towards understanding metallic hydrogen itself,” he told physicsworld.com. The team, which also includes researchers from Henri Poincaré University in Vandoeuvre-lès-Nancy and the University of Rennes and University of Strasbourg, now plans to focus more specifically on superconductivity in silane.



