“You can observe a lot by watching,” opened physicist and former US Secretary of Energy Steven Chu, at his lecture at the 68th Lindau Nobel Laureate meeting in Germany last week. The quote, by baseball player Yogi Berra, is a nod to the biomolecular and biomedical imaging research that the 1997 physics Laureate now focuses on. Chu presented several new, as yet unpublished findings in the lecture in Lindau.
Based at Stanford University, a major focus of Chu’s lab is rare earth nanoparticle biomolecular probes. Attached to proteins in live cells, these probes emit light when optically stimulated, enabling the molecules to be imaged at high resolution using optical microscopy. Their main application is in basic biology research.
Lighting proteins more brightly
In 2010, Chu’s lab began to develop nanoparticles, after the ones they were using hit a ceiling in brightness and therefore imaging sensitivity. Their new design comprised a 5 nm pure crystal core of sodium yttrium fluoride (NaYF4), surrounded by a shell of the same material, but doped with ytterbium and erbium ions. These, in turn, were enclosed in an inert 28 nm-diameter outer shell. The ytterbium–erbium pairing generates upconversion, where illumination with infrared photons stimulates the emission of shorter wavelength visible light for imaging.
The core-shell-shell structure enabled significantly greater ytterbium doping compared with previously reported nanoparticles comprising a simple doped spheroid. This, in turn, increased upconversion, resulting in dramatically brighter nanoparticles.
In the first tests of the nanoparticles this year, an illumination intensity of 8 W/cm2 resulted in luminescence 150 times brighter than the simple spheroid nanoparticles. These were the first images of an individual nanoparticle of that size using an illumination intensity less than 1 kW. At the sub-watt illumination intensities used in in vivo animal studies, the difference becomes greater still. Here, the researchers estimate that the new nanoparticles are three orders of magnitude brighter.
Tracking proteins in live cells
A valuable application of the rare earth nanoparticles for biologists is the tracking of proteins in live cells as a means to uncover their inner workings.
In a spectacular example also demonstrated by Chu’s lab this year, the nanoparticles were used to track the transport of nerve growth factor along the axon of a live dorsal root ganglion neuron at room temperature. Imaging with a frame rate of 2.5 ms and spatial resolution of 2 nm, even the individual steps taken by the molecules could be discerned.
Consequently, transport to and from the neuron body could be seen. The capability is a valuable one, as retrograde transport has been implicated in neurodegenerative conditions such as Alzheimer’s disease.
Photostability is an important advantage of the rare earth nanoparticles in tracking experiments. Organic dyes, for example, photobleach in seconds, while the rare earth nanoparticles are fully photostable and continue to luminesce for hour to days, enabling longer observation. Unlike quantum dots, the particles also do not blink, enabling uninterrupted observation. In an additional benefit, the particles are non-toxic.
Future applications will include investigations of immune and cancer cell behaviour and the differentiation of stem cells in animals, predicts Chu.
Guiding tumour surgery
The brightly emitting nanoparticles could also benefit patients directly. In new research, Chu’s group is collaborating with surgeons at Stanford on their real-time use during tumour excision. Traditionally, surgeons rely on visual inspections and touch to identify tumour margins, which can result in incomplete excision.
Instead, the collaboration plan to use the nanoparticles combined with cancer-specific antibodies to target and light up the tumour when excited by low-intensity infrared light. Their goal is for the surgeon to eyeball the tumour margins, without a camera, exploiting the intense luminescence of the nanoparticles.
New approaches to ultrasound imaging
Highlighting a second major area of investigation, Chu described several ongoing projects in ultrasound imaging, the findings of which are still to be published.
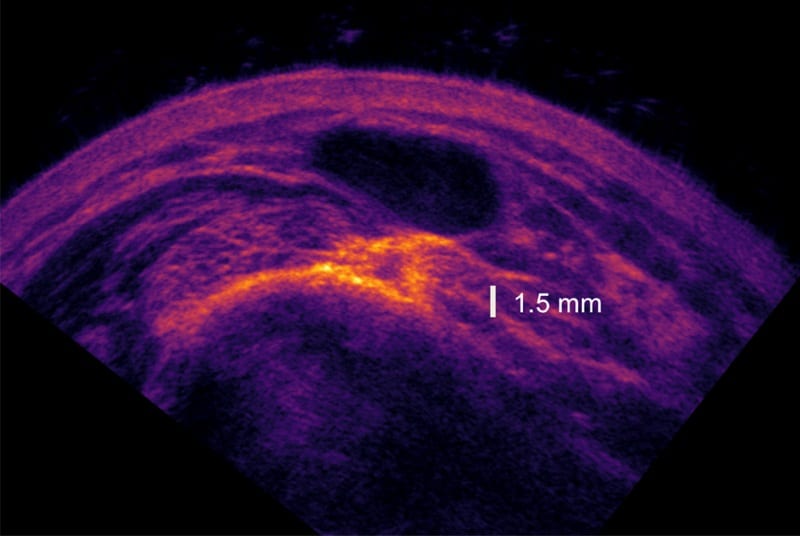
Speckle is a classic feature of traditional ultrasound images and compound imaging is a well-established strategy to remove it, spatially, by acquiring images from several angles or spectrally using more than one frequency.
In a new step, Chu’s group has combined both types of compounding. In doing so, they demonstrated overall reductions in speckle that are the multiple of improvements provided by the techniques individually. In an example of a wrist in vivo, a six- to eight-fold reduction over conventional ultrasound was obtained using angular compounding with nine angles and spectral compounding using two frequencies. Spectral compounding alone provided only a two- to three-fold reduction.
The researchers have also extended their technique with an algorithm to correct distortions of the scan volume due to patient motion, transducer pressure and weak acoustic lensing effects by the tissue. To do so, they exploited computational methods developed for convolutional neural networks combined with NVIDIA GPU chips that “mapped beautifully” on to the problem.
Currently, the new techniques take around 10 s, but with advances in GPUs, computational times will drop below one second, predicts Chu. At a frequency of 15 MHz, the approach produces diffraction-limited images with a transverse spatial resolution of approximately 90 µm.
Chu’s lab is also developing a nonlinear ultrasound technique that promises significantly greater image contrast than conventional imaging. The approach exploits a difference-frequency of 1 MHz generated when tissue is simultaneously insonated at 5 and 6 MHz.
Potential applications include tumour imaging. In an example, Chu showed preliminary data of a nonlinear image of a glioblastoma brain tumour in a mouse. It showed bright spots matching the location of the tumour, verified by pathology, which were not discernible in a conventional image.
Steven Chu’s full lecture – Recent Advances in Biomolecular and Biomedical Imaging – can be seen below. (Courtesy: Lindau Nobel Laureate meetings)