
Stereotactic body radiation therapy (SBRT), which delivers an ablative radiation dose in a few fractions, provides a potent treatment option for patients with liver metastases. To minimize normal tissue damage, SBRT requires highly conformal dose delivery, making it essential to compensate for respiration-induced liver motion.
One option is to perform treatment planning and patient positioning based on the mid-position CT, which represents the time-averaged tumour position, and then add margins to account for uncertainties. This approach, however, relies on a stable mid-position. Any baseline drifts during radiation delivery will change this mid-position with respect to the treatment beam.
Researchers from the Netherlands Cancer Institute (NKI) propose to compensate for such baseline drifts by continuously adjusting the beam aperture according to the motion of the tumour mid-position — a technique called tumour trailing. They investigated whether the use of tumour trailing on the Elekta Unity MR-Linac could provide superior target coverage to non-adapted treatments (Int. J. Radiat. Oncol. Biol. Phys. 10.1016/j.ijrobp.2018.09.011).
The MR-Linac provides real-time monitoring of intra-fractional tumour motion using cine-MRI. “This ability to monitor the tumour is a crucial enabler for trailing,” explains first author Martin Fast. “By addressing the systematic baseline motion, tumour trailing ideally complements mid-position-based treatments or gating approaches that excel at mitigating periodic motion.”
Fast notes that the NKI began clinical treatments on its MR-Linac last month. “While we started with prostate and rectal cancer, we are actively preparing to roll-out MR-Linac treatments for oligometastatic liver patients,” he says.
Treatment simulations
Fast and colleagues began by simulating tumour trailing delivery of liver SBRT, for 17 patients with oligometastatic liver disease previously treated on a conventional linac. They replanned all cases for MR-Linac delivery and simulated treatments in silico using a dedicated machine emulator. Tumour trailing was performed by adjusting the segment apertures according to the averaged tumour position (measured using cine-MRI) over the preceding three respiratory cycles.
The researchers chose to average position measurements from three cycles to balance the trailing lag against accuracy. They determined that imaging the tumour at a frequency of 1 Hz was sufficient to combat motion detection uncertainties: for a respiratory period of 4 s, the effective trailing lag was 6.5 s and the resulting beam-to-mid-position misalignment (for drifts of up to 10 mm) was less than 1 mm.
The simulations modelled respiratory motion as a superposition of periodic motion (with a patient-specific amplitude) and one of five baseline motion scenarios: no drift; continuous linear drift; a single baseline shift halfway through treatment; periodic drift; and MRI-measured baseline drift from three volunteers. The researchers simulated scenarios with and without tumour trailing, and calculated the resulting dose distributions.
Results showed that tumour trailing on the MR-Linac mitigated the dosimetric impact of baseline motion. For the median GTV D98% (dose received by 98% of the gross tumour volume), for example, trailing led to dose increases of 1.9 Gy (linear drift), 1.2 Gy (single shift), 0.7 Gy (periodic drift), and 1.5, 0.8 and 0.5 Gy (for the three measured drifts) per fraction compared with conventional delivery.
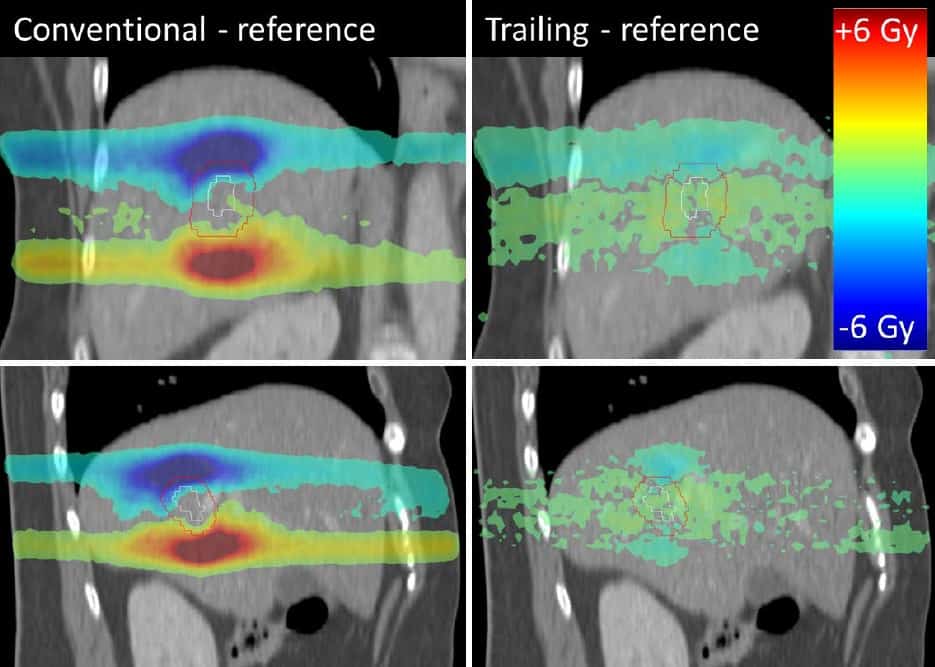
Comparisons with the reference no-drift scenario revealed that tumour trailing restored the dose distributions to those achieved without baseline motion. With trailing, the median residual dose deviations were no greater than 0.7 Gy per fraction.
Experimental validation
To validate tumour trailing on the MR-Linac experimentally, the researchers used a programmable motion phantom to perform continuous periodic motion (4 s period, 15 mm peak-to-peak CC amplitude) along with a linear drift. The phantom insert contained a spherical target and two layers of Gafchromic EBT3 film. They delivered the treatments established in the in silico study to the phantom, and used gamma tests to compare films irradiated using conventional or trailing delivery with films irradiated in the reference scenario.

The phantom experiment confirmed the predicted dosimetric gains. Compared with conventional delivery, tumour trailing increased the local gamma pass rate from 40±3% to 82±19% (3%/1 mm), 49±3% to 98±3% (3%/2 mm) and 58±4% to 99±1% (3%/3 mm).
The authors concluded that tumour trailing offers a robust way to compensate for baseline motion during MR-Linac treatments, without increasing delivery time. “Deploying tumour trailing clinically is on our roadmap for 2019,” Fast tells Physics World. “Prior to the clinical introduction, several technical and logistical hurdles such as implementing effective online quality assurance procedures need to be overcome.”



