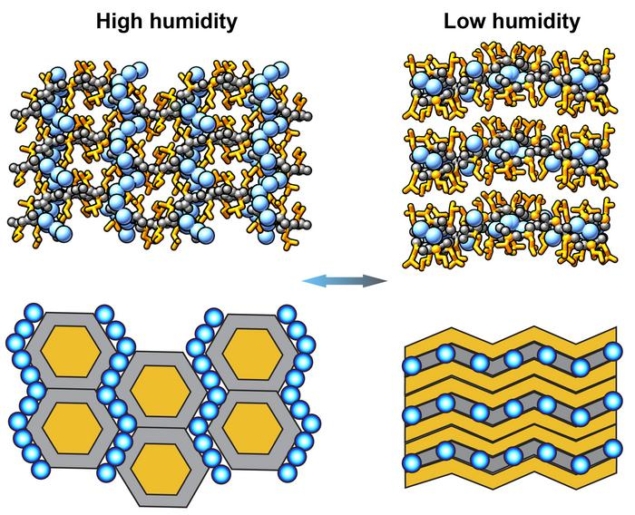
Protein molecules are highly dynamic, continually changing shape in response to changes in external conditions. Scientists have long sought to mimic this behaviour in artificial materials, and now a team at the City University of New York (CUNY) in the US has done just that, constructing a crystalline solid that switches between several distinct architectures as the ambient humidity changes. Their work could make it easier to fabricate adaptive materials on a large scale for applications such as humidity-responsive coatings.
Proteins owe their shape-shifting character to a series of complex interactions that take place between two or more molecules. These supramolecular interactions, as they are known, allow proteins to adapt their properties – and therefore their functions – as needed. Water plays an important role in such interactions because it stabilizes certain structures while weakening others.
“Stripped-down” versions of protein behaviour
In the new work, researchers led by CUNY chemist Rein Ulijn and chemical engineer Xi Chen studied peptides, which are the molecular building blocks that make up proteins. In particular, they focused on leucine (L) and isoleucine (I), which are isomers, meaning they have the same chemical formula but different structures. “Such short peptides give us access to ‘stripped-down’ versions of protein behaviour,” explains Ulijn, who is also the founding director of CUNY ASRC Nanoscience Initiative. “They’re simple enough to design systematically, but still rich enough to encode sometimes surprisingly complex and dynamic behaviour.”
They found that when the chemical potential of water in the system – effectively, the humidity – changed, the solid-state porous architecture of LI crystals reorganized, reversibly switching between rigid perpendicular/parallel honeycomb structures and layered soft van der Walls structures. Importantly, Ulijn explains, this transformation occurs without compromising the peptides’ overall structural integrity.
“What makes this particularly significant is that most dynamic supramolecular systems are limited to relatively minor changes in organization,” he says. “In contrast, the peptide side chains in our system undergo very dramatic conformational reorganization, which translates into the topological changes observed.”
Uljin adds that this process offers a completely new way to design materials that can switch between distinct structural states. “This opens the door to solid materials that are both robust and highly adaptable, a combination that is difficult to achieve with existing approaches,” he tells Physics World.
A new toolbox for designing dynamic solid-state materials
The researchers say they undertook their study to address a “fundamental gap between biological systems and synthetic solid-state materials”. Although proteins routinely undergo sequence-encoded conformational changes to access multiple functional states in solution, replicating this kind of dynamic behaviour in solid materials has been a major challenge. “Our goal was to create a minimalist, peptide-based system that could mimic this adaptability without relying on large, complex structures and that could be triggered by low energy inputs,” they explain.
Cross-linked polymer is both stiff and stretchy
The team says the work provides a new toolbox for designing dynamic solid-state materials with tuneable topology and function, which could potentially impact a wide range of fields. One potential application is the development of adaptive materials with switchable mechanical properties, where stiffness and softness can be controlled through environmental humidity or temperature. “This could be useful in soft robotics, responsive coatings, or smart structural materials,” Chen notes.
The researchers are now studying other peptide structures in hopes of better understanding the fundamental rules for conformational control of short peptides. Ultimately, they say this programme should lead to specific design rules for porous peptide materials, making it possible to explore a broader range of sequences and side-chain chemistries. “We are also interested in scaling these materials to enable practical demonstrations in hydration-responsive coatings,” Chen adds.
The team reports its work in Matter.




