 Read article: Physics meets fashion as bioluminescent dress debuts at Paris Haute Couture Week
Read article: Physics meets fashion as bioluminescent dress debuts at Paris Haute Couture Week
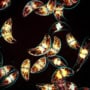
Physics meets fashion as bioluminescent dress debuts at Paris Haute Couture Week
The spectacular “living” garment was part of Iris van Herpen’s new fashion collection – Sympoiesis








