Physical chemists in China have made carbon-50 molecules in the solid state for the first time. Lan-Sun Zheng and colleagues at Xiamen University, and co-workers at the Chinese Academy of Sciences in Beijing and Wuhan, prepared the molecules – which they describe as a long sought little sister of carbon-60 – in an arc-discharge technique involving chlorine. The result will allow scientists to study the properties of carbon-50 with a view to exploiting its unusual properties. The method developed by the Chinese team also opens the way to making other small, cage-like carbon molecules or "fullerenes" (S-Y Xie et al. 2004 Science 304 699).
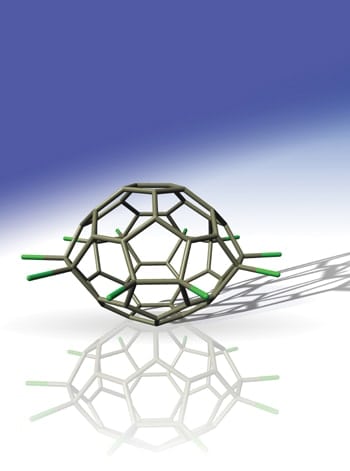
The most common fullerene is carbon-60 – also known as buckminsterfullerene or “buckyball”. This molecule, which contains 60 carbon atoms arranged in a spherical structure made up of pentagons and hexagons, was first created in 1985. Since then larger fullerenes containing between 70 and 500 carbon atoms have also been produced.
All the fullerenes made so far obey the isolated pentagon rule (IPR): this rule states that the most stable molecules are those in which every pentagon is surrounded by five hexagons. However, it is not possible to satisfy this rule in a molecule with fewer than 60 carbon atoms. This means that so-called non-IPR fullerenes should have unusual properties, but it also makes them structurally unstable and difficult to synthesise. Until now, fullerenes with fewer than 60 carbon atoms have only ever been made in the gas phase.
Zheng and colleagues succeeded in stabilising and capturing solid-state carbon-50 molecules using a graphite arc-discharge method. They added 0.013 atmospheres of carbon tetrachloride vapour to 0.395 atmospheres of helium in a sealed stainless steel vessel and then applied an electric field of 24 Volts. After purifying around 90 grams of soot that contained carbon-50 chloride (C50Cl10), they obtained about 2 milligrams of C50Cl10 that was 99.5% pure.
“The C50Cl10 looks like a spacecraft or a spinning planet with 10 reactive carbon-chlorine arms ready for further chemical functionalization,” team member Su-Yuan Xie told PhysicsWeb (see figure). Like derivatives of carbon-60 and 70, Xie says that carbon-50 could easily react with a variety of organic groups to form new compounds with interesting chemical and physical properties. Moreover, the technique could also be extended to synthesise other small fullerenes, such as carbon-54 and carbon-56.



