If you think you have seen the above headline somewhere before, then you probably have. In 1999, nuclear physicists at Lawrence Berkeley National Laboratory in the US claimed to have produced three atoms of the super-heavy element with atomic number 118 -- the heaviest ever detected. But that claim was retracted three years later when it emerged that some of the data had been falsified.
This time, however, element 118 — and its slightly lighter counterpart element 116 — is here to stay, pushing back the boundaries of the periodic table and helping researchers to understand why some nuclei are more stable than others (Phys Rev C 74 044602).
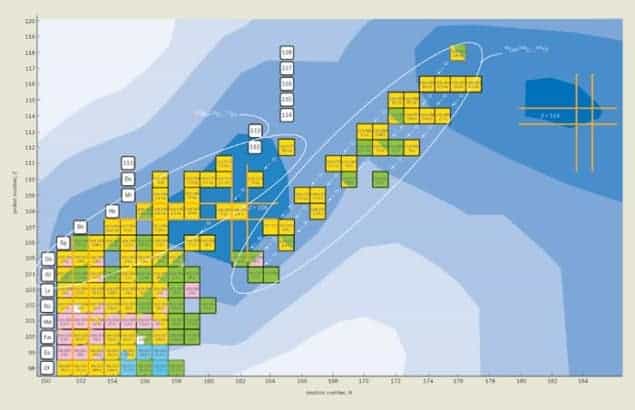
The world is made up of about 90 naturally occurring elements, but since the 1940s physicists have been able to produce heavier, less stable elements. Such elements are vital for testing models of the nucleus, and to date researchers have amassed data on 29 “super-heavy” nuclei with atomic numbers between 104 and 118.
The definitive discovery of element 118, which is expected to be a noble gas that lies right below radon in the periodic table, was a collaborative effort between researchers at the Livermore lab and the Joint Institute for Nuclear Research (JINR) in Dubna, Russia. Based on data taken early last year in experiments at the JINR cyclotron, in which a target of californium (atomic number 98) is bombarded with a beam of calcium-48 ions, the team observed alpha decay chains that could only occur if element 118 exists. In these decay chains, previously observed element 116 is produced via the alpha decay of element 118.
“Element 118 is the last one in a series of superheavy elements produced in nuclear reactions with calcium-48,” says Dubna team-leader Yuri Oganessian. “So far all the experimental data point to the existence of an ‘island of stability’ in the region of superheavy elements, as theory says it should.”
As well as providing a clear ending to a rather unsavory chapter in physics, the discovery of element 118 brings to five the number of new superheavy elements discovered by the Livermore–Dubna collaboration (113, 114, 115, 116 and 118). Next year the team plans to look for element 120 by bombarding a plutonium target with iron isotopes.