A firm in the US is drawing up plans for a badge–sized, wearable sensor that can detect in real time the presence of E. coli, anthrax, salmonella and other biological threats. The sensor, which contains tiny diamond cantilevers, is being developed by Advanced Diamond Technologies (ADT) in Illinois. The company is currently six months into a three-year research programme and hopes to have prototype devices available by the end of 2011.
Diamond is well known for being exceptionally hard and a good conductor of heat. But it also has other properties that make it useful as a biosensor. In particular, the surface of diamond is covered with strong hydrogen–carbon bonds, which means that it is stable in water, unlike other sensor materials like silicon. Moreover, the hydrogen atoms can be stripped off and replaced with antibody molecules that can bond, like a lock and key, with a target biomolecule like E. coli.
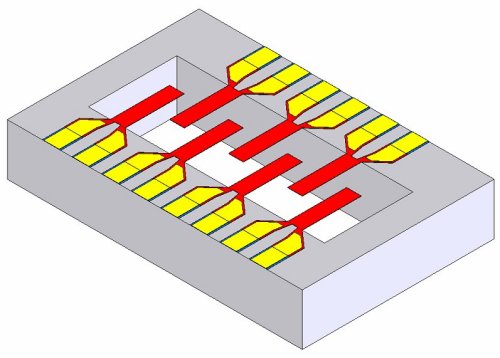
The new device consists of diving–board-shaped cantilevers, each about 100 μm long, mounted on a semiconductor chip. Each diamond cantilever is highly uniform and consists of nanocrystalline grains, each about 2–5 nm in diameter, deposited using chemical–vapour deposition. Any biomolecule landing on the surface of the cantilever changes the device’s vibrational frequency, which can be converted into an electrical signal through the piezoelectric response of the cantilever.
Detecting 100 cells
To ensure that the signal is strong enough, ADT is planning to incorporate as many as 50 individual cantilevers in each sensor. One challenge will be to concentrate the pathogenic agents so that even tiny amounts can be detected — the initial target is to detect 100 cells in 100 μl of fluid. The sensor could even be used to detect a range of different target molecules by simply attaching different antibodies to each cantilever.
“We want to miniaturize the sensor so that it can be worn as a badge or around the neck,” says lead investigator John Carlisle. The final device will also have to communicate its signal wirelessly so that, say, a firefighter wearing the sensor is aware of potentially hazardous conditions in a building and also that information is sent to a centralized response team.
Although the project is being fully funded by a $4.8m contract from the US Defense Threat Reduction Agency, the firm says the sensor could have non-military applications such as determining whether, say, water is safe to drink. Carlisle even wants to adapt the sensor so that it can detect not just water–based biomolecules but those that are air-borne too. “There are very sizeable opportunities,” he says.



