Researchers in the US have invented a “lock and key” technique that causes small particles to assemble themselves into a variety of tiny structures. The method could offer a simple way to create technologically useful materials on the micrometre and nanometre length scales.
Particles measuring between 100 nm and 1 µm are excellent building blocks for making optoelectronic devices. Such particles are about the same size as the wavelengths in the visible part of the electromagnetic spectrum and so interact strongly with light.
One promising way of creating devices is to disperse the particles in a liquid to form a colloid that can then solidify to create a colloidal crystal. The optical properties of such crystals can be tuned by changing the spacing between particles. Devices are usually created by exploiting the ability of some particles to self-assemble into specific structures. Surface chemistry can be used to control the shapes of these structures by coating the particles with molecules, such as DNA strands, that bind to each other.
Shaping a few basic building blocks
Now, Stefano Sacanna and colleagues at New York University have invented a new control method that does not depend on the surface chemistry of particles, but only on their shapes. As a result, the process avoids the many problems associated with coating particles with molecules or treating their surfaces. “Ideally, you could design a cluster with precise geometry and well-defined physical and chemical properties by shaping a few basic building blocks and letting them self-assemble with this simple ‘lock and key’ mechanism,” says Sacanna.
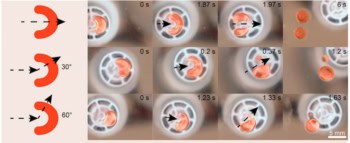
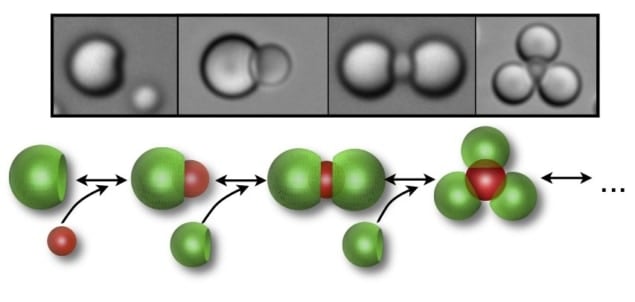
The technique employs two particles with complementary features – for example a spherical cavity on one particle (the lock) and a matching spherical protrusion on the other (the key). The particles are pushed together by the “depletion interaction”, which involves the addition of a third type of particle that is much smaller than either the lock or key. When a lock and key come very close together, the small particles can no longer fit between them. Because there are no small particles between the large particles to push them apart, the particles start to close in on each other as if they are attracted by a short-range force.
The strength of the depletion interaction is proportional to how good the fit is between the lock and key – if the fit is poor, some tiny particles can get into the gaps and push the lock and key apart. As a result, the particles tend to form structures in which locks and keys are stuck together (see figure).
Reversible process
Thanks to the fact that the technique does not depend on controlling the surface chemistry of particles, it allows for more freedom in designing and assembling functional clusters, claims Sacanna. The absence of chemical bonds also means that assembly is reversible. Clusters can be taken apart by simple changing the temperature.
This novel and essential feature can be used to create, for example, mobile parts in micromachinery Stefano Sacanna, New York University
Another important benefit of the technique is that lock-and-key bonds are much more flexible than chemical bonds. “This novel and essential feature can be used to create, for example, mobile parts in micromachinery,” Sacanna tells physicsworld.com.
Several locks may bind to a single key (see figure), which means that a structure can have a number of bonds. It may also be possible to create more than one “pocket” on a lock particle. According to Michael Solomon of the University of Michigan, multiple pockets “would introduce the colloidal equivalent of extended co-ordination complexes – 2D and 3D molecular arrays that self-assemble in fixed geometries from atom and ligand molecules”. “The assembly of colloidal particles might allow access to desirable, but so far elusive, complex colloidal structures,” he adds.
Sacanna and colleagues now plan to make “smart” particles and push their self-assembly to the limit where well-defined structured clusters of particles can self-replicate.
The work is described in Nature 464 575.