Researchers at Stanford University in the US have found a way to generate light deep within living tissues, potentially leading to new forms of gene and cancer therapies. The proof-of-concept approach uses ultrasound to trigger luminescence in nanoscale particles travelling through the bloodstream, and it has already been tested in tissue-mimicking “phantoms” and live mice. However, its developers caution that human trials are still some way off.
Light has numerous applications in medicine and biological research. It is widely used, for example, to stimulate cell growth and in photodynamic therapies for skin and eye conditions, as well as certain types of cancer.
The problem is that many potentially useful wavelengths of light are easily scattered by tissues and become attenuated over relatively short distances. This means they cannot penetrate very far into the body without help from invasive methods such as removing overlying tissue or inserting/injecting optical implants and light-emitting nanoparticles into the target area.
Sound and light
The new work by Stanford materials scientist and engineer Guosong Hong and colleagues involves nanoparticles made from a ceramic material with the chemical formula Sr4Al14O25:Eu,Dy. This material is mechanoluminescent, meaning that it emits light when subjected to mechanical stresses and deformations. In Sr4Al14O25:Eu,Dy, these mechanoluminescent effects can be induced by exposing the material to sound waves, which penetrate more deeply into tissue than light waves.
The Stanford researchers began by coating their nanoparticles with a biocompatible film. They then suspended the particles in a solution and injected the resulting colloid into the veins of mice. Thanks to the rodents’ vascular systems, the particles soon travelled to all parts of their bodies.
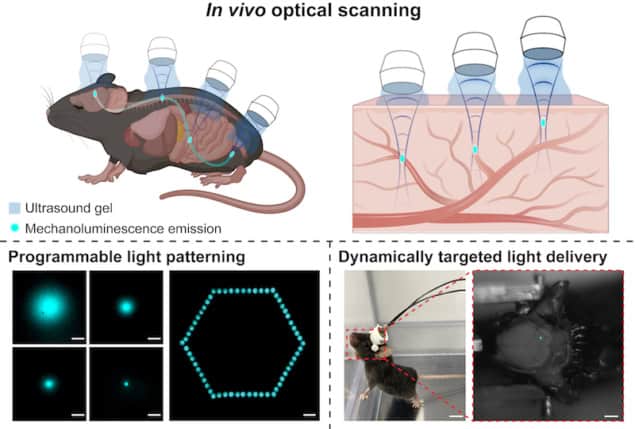
The researchers then showed they could make the nanoparticles emit blue light with a wavelength of 490 nm simultaneously in multiple locations (such as the brain, gut, hindlimb and spine) by applying sound waves to different parts of the mouse’s body. In addition, they showed they could create precise patterns of in-situ light generation throughout the three-dimensional volume of the animal, controlled over distances of 100 to 200-μm in the focal region. The ultrasound can also be used as a scanner to define where the light is generated.
A host of applications
The team picked the 490 nm wavelength because it has many applications, including neuron modulation and photodynamic cancer therapy. However, applying the same technique to different materials could produce other useful wavelengths, too. Indeed, Hong and his colleagues are exploring the possibility of using materials that emit ultraviolet light, which has antiviral and antibacterial properties.
The researchers say their approach is broadly applicable to virtually all therapeutic modalities that requires light to be delivered deep within the body, including optogenetics, phototherapy and photo-switchable gene editing. This last technique currently suffers from off-target effects, but the researchers say that by pairing light-producing nanoparticles with a light-activated gene-editing system, they may be able to use ultrasound to turn gene editing on and off in localized areas of the body.
“The overarching theme of my lab’s research is to develop new strategies to deliver and receive light throughout the body in its native, living state,” Hong tells Physics World. “In 2024, we reported on a method to render living tissue transparent using strongly absorbing dye molecules. In the present study we have taken a complementary approach: rather than modifying how light propagates through tissue, we leverage the intrinsic penetrative capability of ultrasound, together with the pervasive reach of the circulatory system, to generate light directly within deep regions of the body.”
Light-absorbing dye turns skin of a live mouse transparent
Reporting their work in Nature Materials, the researchers are now working to integrate their approach with other light-activatable control systems, including photo-switchable Cas9 gene editing in collaboration with Michael Lin’s lab at Stanford. In parallel, they hope to develop alternative mechanoluminescent materials that will break down safely in the body. While the materials studied in this work did not seem to show adverse effects in mice, they also did not break down quickly, and the researchers say they could accumulate in organs such as the liver.
“What we’re demonstrating here is a proof-of-concept showing that you can produce light emission in a programmable manner deep within the body,” Hong says. “If we can replace the material with one that is safer to be used in humans, that will start to pave the way for clinical applications.”