The amount of oxygen that a heart consumes is a key indicator of its health. If the heart is not receiving or using enough oxygen, heart tissue can be damaged, contributing to future heart failure.
With abnormal myocardial oxygen consumption an indicator of potential cardiac dysfunction, its measurement could help in the early detection and treatment of heart failure. And as one in four individuals are likely to develop heart failure in their lifetime, this is of critical importance. But measurement of myocardial oxygen consumption is not a simple process. The gold standard for determining the heart’s oxygen use is cardiac catheterization. But this test – which involves threading a catheter from a patient’s neck or groin into the coronary sinus (CS), the largest coronary vein – is highly invasive, time-consuming and comes with a level of risk.
A new MRI technique may soon offer a rapid, non-invasive alternative. Developed by an international research team headed up at Cedars-Sinai Health Sciences University, the high-resolution MRI method can assess the heart’s oxygen consumption in just three minutes. In an initial study of 22 patients with heart failure, reported in Science Translational Medicine, the team validated its accuracy, feasibility, performance and repeatability.

MRI is sensitive to blood oxygenation via the blood oxygen level–dependent (BOLD) signal, originally developed for mapping brain activity. Use in the heart remains challenging, however, due to the need for complex calibration, motion sensitivity and long acquisition times. Hsin-Jung Yang, of the Biomedical Imaging Research Institute at Cedars-Sinai, and collaborators overcame these obstacles by developing a rapid, self-calibrated cardiac MRI framework that enables free-breathing blood oximetry (measurement of blood oxygen saturation) in the CS and quantification of whole-heart myocardial oxygen extraction, without requiring contrast agents or pharmaceutical stress.
The researchers’ primary objective was to determine the accuracy and precision of MRI-derived measurements of CS blood oxygenation, compared with those obtained by invasive CS catheterization. They also aimed to perform non-invasive quantification of global myocardial oxygen consumption and myocardial oxygen efficiency, with comparisons between healthy controls and patients with heart failure.
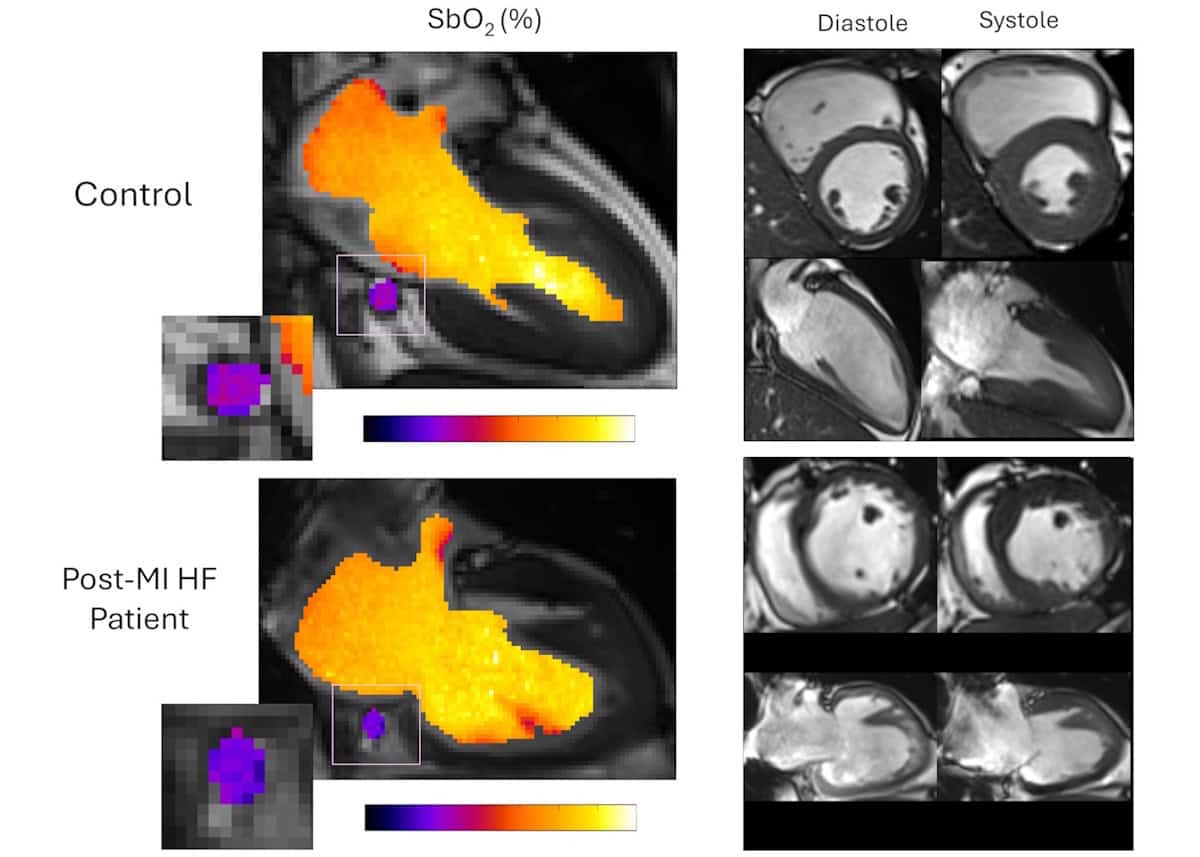
To achieve this, they developed a motion-resolved reconstruction algorithm for cardiac BOLD MRI that enables clear imaging of the moving heart during breathing and heartbeats. The team first validated the method in pigs, and then applied it to a group of 22 patients with heart failure and a history of previous heart attack, as well as 11 healthy volunteers.
The researchers acquired clinical cine images to define the cardiac anatomy, localize the CS and measure ventricular function for estimating the oxygen–mechanical work coupling efficiency. Using this approach, they identified impaired myocardial oxygen consumption in the patient group, including those with preserved ejection fraction (how much blood the left ventricle pumps out with each contraction, a low value of which can indicate a heart problem). The finding that impaired oxygen consumption was measurable even before detectable structural or functional decline may facilitate the early detection of cardiac dysfunction.
The researchers note that their self-calibrated MRI framework directly addresses the difficulty of performing quantitative oximetry of the CS – a mobile blood vessel that undergoes marked displacement throughout the cardiac cycle. “Our framework directly addresses these challenges with a continuous, free-breathing, motion-resolved 3D acquisition that retrospectively sorts data across cardiac and respiratory phases, ensuring stable CS tracking despite its complex motion and size variation,” they write.
By eliminating the dependence on gating and calibration, the method could be applied across diverse clinical populations, including those with arrhythmias, intolerance of breath-holding or physiologic stress, for whom conventional gated acquisitions are unreliable. The team suggests that the framework also holds promise for extending oxygen consumption imaging to other moving organs, such as the liver and kidney, and that in the future, the motion-resolved BOLD framework could be applied to tissue-based quantification.
The heart of the matter: how advances in medical physics impact cardiology
The researchers are performing ongoing clinical studies to evaluate the MRI technique in aortic stenosis (narrowing of the aortic valve) and hypertrophic cardiomyopathy (thickening of the heart muscle), where altered oxygen extraction and metabolic efficiency have revealed disease severity, risk and treatment response beyond conventional imaging.
More broadly, the Yang Lab is extending this approach to characterize oxygen utilization in all cardiometabolic diseases and associated emergent therapies, with the goal of noninvasively defining myocardial energetic supply–demand balance, identifying therapy–response phenotypes, and monitoring disease progression and metabolic remodelling over time.
“By enabling a fast, non-contrast, non-ionizing radioactive method for measuring cardiac oxygen metabolism, [this MRI method] can unlock frontiers for early diagnosis, personalized therapy, and the development of next-generation cardiometabolic treatments to combat the global heart failure epidemic,” the team concludes.




