Glasses are much more mysterious than their crystalline counterparts, yet have a wealth of practical uses, says Jon Cartwright
At the British Museum in London, there is a small turquoise-blue jug, originating from Egypt under the reign of the pharaoh Thutmose III. About the size of a salt shaker, the pretty opaque object was probably designed to hold perfumed oil, and is made almost entirely of glass. Yet despite being over 3400 years old, it is not considered one of the earliest examples of human glass making. Historians believe that Mesopotamians were amongst the leading glass-making cultures, fashioning beads and other simple decorative items from glass as long as 4500 years ago.
At first glance, glass does not seem very complicated. It merely refers to a material that has an amorphous rather than a crystalline structure – that is, one in which the atoms or molecules have no long-range order. Almost all common glasses, including those made by the ancient Egyptians and Mesopotamians, involve melting just three ingredients: silica (sand) for the basic structure; along with an alkali oxide (typically soda, or sodium carbonate) to lower the melting temperature; and lastly, calcium oxide (lime) to prevent the mixture from being soluble in water. In fact, the recipe can be simpler still, for we now know that almost any material can turn glassy if it is cooled from its liquid state so fast that its atoms or molecules are arrested before they have a chance to form a well-ordered solid state. But this simple description belies the depth of physics going on under the surface – physics that has been the subject of intense research for well over a century, with some aspects that still baffle us today.
The biggest question physicists want to answer is why a cooling liquid forms a hard glass at all, when no distinct change in structure occurs between the liquid and glass states. One might well expect glass to deform like a very viscous liquid. Indeed, there is a persistent myth that glass in old window panes is warped because it flows slowly over time (see box “The flowing myth”). In truth, glass is hard and brittle, and remains stable over surprisingly long periods. The stability of glass is one of its most attractive characteristics, for example in the storage of nuclear waste.
An ideal glass is where molecules are packed together in the densest possible random arrangement
As seen through the conventional lens of “phase transitions”, put forth by Soviet physicist Lev Landau, there is no sudden shift in the underlying order (at least, no obvious one) when a substance turns into a glass – as would be seen for the emergence of any other genuine state of matter. The main difference between a liquid and a glass is that a liquid can continue to explore different disordered configurations, whereas a glass is, more or less, stuck with one. What makes a cooling liquid select a particular state on transition to glass is a question that goes back over 70 years (see box “In search of the ‘ideal’ glass”).
The fact that, as an amorphous solid, a material can potentially adopt so many different states makes glass incredibly versatile. With small changes in composition or processing, glass properties vary wildly (see box “Two routes to better glass”). This accounts for the huge range in glass applications – from camera lenses to cookware, from windscreens to staircases, and from radiation protection to fibre-optic cables. Smartphones too, as we know them, would not have been possible without the development of thin-but-strong glass, such as “Gorilla Glass” glass, first made by the US manufacturer Corning. Even metals can turn into glass (see box “Mastering the metallic”). Often, the optical and electronic properties of a material do not differ greatly between its glassy and crystalline states. But sometimes they do, as is seen in phase-change materials, which, besides being of importance for data storage, are offering fundamentally new insights into chemical bonding (see box “The future of phase-change materials”).
Perhaps the most surprising question to ask about glass is not what it is, but what it isn’t
However, perhaps the most surprising question to ask about glass is not what it is, but what it isn’t. While we are accustomed to thinking of glass as a hard, transparent substance, a vast swathe of other systems exhibit “glass physics”, from ant colonies to traffic jams (see box “Glass where you least expect it”). Glass physics helps scientists to understand these analogues, which in turn can shed light on glass physics itself.
The flowing myth

Look through the stained-glass windows of any medieval church, and you will almost certainly see a distorted view. The effect has long led scientists and non-scientists alike to suspect that, given enough time, glass flows like an exceptionally viscous liquid. But is there any validity to this claim?
The question is not as straightforward as it might at first appear. In truth, no-one can say precisely when a liquid stops being a liquid and starts being a glass. Conventionally, physicists say a liquid has become a glass when the atomic relaxation – the time for an atom or molecule to move a significant portion of its diameter – is longer than 100 seconds. This relaxation rate is about 1010 times slower than in runny honey, and 1014 times slower than in water. But the choice of this threshold is arbitrary: it reflects no distinct change in the fundamental physics.
Even so, a 100 second relaxation is definitive for all human purposes. At this rate, a piece of common soda-lime glass would take aeons to slowly flow and turn into the more energetically favourable crystalline silicon dioxide – otherwise known as quartz. If the stained glass in medieval churches is warped, therefore, it is more likely a result of the original glassmaker’s (by modern standards) poor technique. On the other hand, no-one has performed a thousand-year experiment to check.
In search of the “ideal” glass

As a liquid cools, it can either harden into a glass, or crystallize. However, the temperature at which a liquid transitions to a glass is not fixed. If a liquid can be cooled so slowly that it does not form a crystal, then the liquid will ultimately transition to a glass at a lower temperature, and form a denser one as a result. The US chemist Walter Kauzmann noted this fact in the late 1940s, and used it to predict the temperature at which a glass would form if a liquid was cooled “in equilibrium” – that is, infinitely slowly. The resultant “ideal glass” would, paradoxically, have the same entropy as a crystal, despite still being amorphous, or disordered. Essentially, an ideal glass is where molecules are packed together in the densest possible random arrangement.
In 2014 physicists including Giorgio Parisi of the Sapienza University of Rome in Italy (who shared the 2021 Nobel Prize for Physics, for his work on “the interplay of disorder and fluctuations in physical systems”) worked out an exact phase diagram for the formation of an ideal glass, in the (mathematically easier) limit of infinite spatial dimensions. Usually, density can be an order parameter to distinguish different states, but in the case of glass and a liquid, the density is roughly the same. Instead, the researchers had to resort to an “overlap” function, which describes the similarity in positions of molecules in different possible amorphous configurations, at the same temperature. They found that when the temperature is less than the Kauzmann temperature, the system is prone to fall into a distinct state with a high overlap: a glass phase.
In three dimensions, or indeed any small finite number of dimensions, the theory of the glass transition is less certain. Some theorists have attempted to describe it thermodynamically, again using the ideal glass concept. Others believe it is a “dynamic” process in which, at progressively lower temperatures, more and more pockets of molecules become arrested, until the entire bulk becomes more glass than not. For a long time, proponents of the two camps have been at loggerheads. In the last couple of years, however, condensed-matter theorist Paddy Royall at ESPCI Paris in France and colleagues claim to have shown how the two approaches can be largely reconciled (J. Chem. Phys. 153 090901). “A lot of the resistance [to agreement] we saw 20 years ago has been lost,” he says.
Two routes to a better glass

To change the properties of glass, you have two basic options: alter its composition, or alter the way in which it is processed. For instance, using borosilicate rather than the common soda and lime makes glass less prone to stress when heated, which is why borosilicate glass is often used in place of pure soda lime for bakeware. To make the glass even more robust, its exterior surface can be cooled more rapidly than its bulk in a “tempering” process, as in Corning’s original Pyrex.
Another of Corning’s innovations, Gorilla Glass for smartphones, has a more complicated recipe of composition and processing to achieve its strong, scratch-resistant properties. An alkali-aluminosilicate material at heart, it is produced in a sheet mid-air in a special fast-quenched “fusion drawn” process, before being immersed in a molten salt solution for additional chemical strengthening.
Typically, the denser a glass is, the stronger it is. In recent years, researchers have discovered that very dense glass can be created by physical vapour deposition, in which a vaporized material is condensed onto a surface in a vacuum. The process allows molecules to find their most efficient packing one at a time, like a game of Tetris.
Mastering the metallic

In 1960 Pol Duwez, a Belgian condensed-matter physicist working at Caltech in California, US, was rapidly cooling molten metals between a pair of cooled rollers – a technique known as splat quenching – when he discovered that the solidified metals had turned glassy. Since then, metallic glasses have enthralled materials scientists, partly because they are so hard to make and partly because of their unusual properties.
With none of the grain boundaries inherent in ordinary crystalline metals, metallic glasses don’t wear easily which is why NASA has tested them for use in lubricant-free gearboxes, seen here, in its space robots. These glasses also resist the absorption of kinetic energy – for example, a ball made of the material will bounce for a weirdly long time. Metallic glasses also have excellent soft magnetic properties, making them attractive for highly efficient transformers, and can be manufactured in intricate shapes, like plastics.
A lot of metals will only turn glassy (if they do so at all) at breathtakingly fast cooling rates – billions of degrees per second or more. For that reason, researchers usually seek alloys that transition more easily, typically by trial and error. In the past few years, however, Ken Kelton at Washington University in St Louis, US, and colleagues have suggested that it is possible to predict the likely glass transition temperature by measuring the shear viscosity and thermal expansion of a liquid metal (Acta Mater. 172 1). Kelton and his team ran a research project on the International Space Station, to study the temperature at which a metal actually becomes glassy, and found that the transition process starts while the metal is still a liquid. By measuring how viscous the liquid is the researchers can now determine whether a glass will form, and what some of its properties will be. Should prediction become commonplace, so too could metallic glasses in commercial devices. In fact, the US tech company Apple has long held a patent for the use of metallic glass on smartphone covers, but has never put it into practice – perhaps due to the difficulty in finding a metallic glass that is economically viable.
The future of phase-change materials

The mechanical properties of glasses and crystals may be different, but usually their optical and electronic properties are fairly similar. To the untrained eye, for instance, normal silicon-dioxide glass looks almost the same as quartz, its crystalline counterpart. But some materials – notably chalcogenides, which include elements from the oxygen group of the periodic table – have optical and electronic properties that are markedly different in their glassy and crystalline states. If these materials also happen to be “bad” glass formers (that is, crystallize when modestly heated) then they serve as so-called phase-change materials.
Most of us will have handled phase-change materials at one time or another: they are the data-storage medium of rewritable DVDs and other optical discs. Insert one of these into a suitable drive, and a laser can switch any bit on the disc between the glassy and crystalline state, representing a binary zero or one. Today, optical discs have been largely supplanted by electronic “flash” memory, which has a greater storage density and no moving parts. Chalcogenide glass is also sometimes used in photonic integrated optical circuits, as pictured here. Phase-change materials have continued to find applications in data storage by the US tech company Intel, and its “Optane” brand of memory, which is fast to access yet non-volatile (it is not erased when the power is switched off). This application remains niche, however.
More profitable, says solid-state theorist Matthias Wuttig at RWTH Aachen University, Germany, is to ask where the phase-change property comes from. Four years ago, he and others proposed a new type of chemical bonding, “metavalent” bonding, to explain its origin. According to Wuttig, metavalent bonding provides some electron delocalization, as in metallic bonding, but with an added electron-sharing character, as in covalent bonding. Unique properties, including phase-changing, result (Adv. Mater. 30 1803777). Not everyone in the field wants to add a new type of bonding to the textbooks, but Wuttig believes the proof will be in the pudding. “The question now is whether [metavalent bonding] has predictive power,” he says. “And we are convinced it has.”
Glass where you least expect it
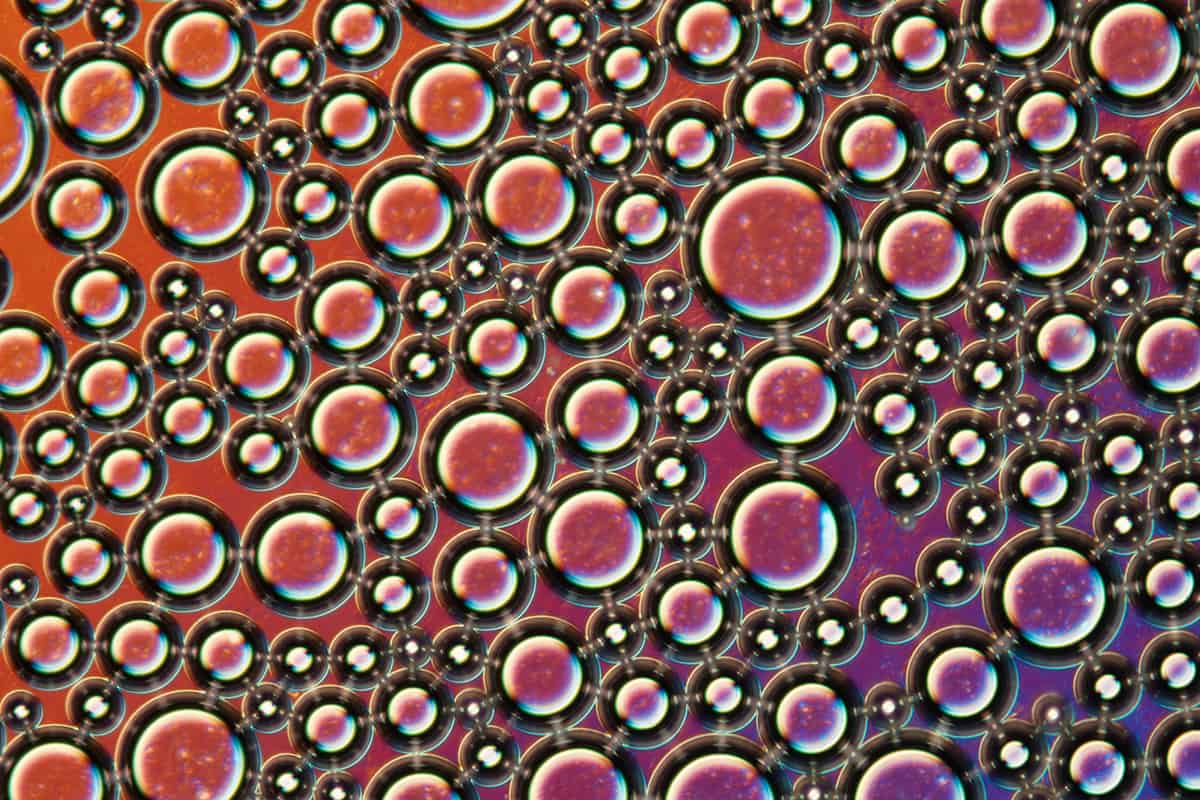
Fans of music festivals will recognize the phenomenon: you are slowly trying to leave a performance along with thousands of other people, when all of a sudden the crowd halts, and you can move no more. Like a molecule in cooling molten silica, your motion is suddenly arrested – you and your fellow festival-goers have turned into a glass. Or a glass analogue, at least.
Other glass analogues include ant colonies, biological cells trapped between slides, and colloids, such as shaving foam (see image above). Colloids in particular, with particles ranging up to microns in size, are convenient systems for testing theories of the glass transition, as their dynamics can actually be seen through a microscope. Even more surprising, though, is the onset of glass behaviour in certain computer algorithms. For instance, if an algorithm is designed to seek out progressively better solutions to a problem with a large number of variables it can become overwhelmed by complexity and grind to a halt before the optimal solution is found. By borrowing statistical methods designed for the fundamental study of glasses, however, such algorithms can be improved, and better solutions found.




