
Researchers in Germany have developed a thermo-responsive hydrogel and loaded it with tendon stem/progenitor cells (TSPCs), developing a therapy for tendon injuries. This hydrogel allows TSPC survival, proliferation and metabolic activity, while being stable and biocompatible (Biomed. Mater. 13 034107).
Tendon injury is common and can be debilitating, leading to deterioration of the patient’s life quality. Current treatments can provide some relief, but with a high recurrence of injury. Long-term solutions require the use of a tendon graft, usually taken from the patient.
Many strategies for tendon regeneration are under investigation and currently involve the use of cells and a scaffold. Two options in the choice of cells can be considered: the first is to implant tendon cells. To harvest those cells, a biopsy from a healthy part of the patient’s tendon is needed. The second option is to use stem cells, which will become tendon cells. Stem cells can be found in almost every tissue, including the tendon (TSPCs). Stem cells have the capacity to generate almost any type of cell, including cells of the bone, cartilage, tendon, brain, liver and more. Their capacity for self-renewal makes them of great interest in research.
In vivo, cells are supported physically, mechanically and biochemically by a scaffold known as the extracellular matrix (ECM). Scaffolds for therapeutic use are designed with dimensions that match the injury to repair. As a consequence, critical injuries would require big scaffolds and thus surgery for implantation. Invasive surgery can be avoided, however, by using an injectable scaffold that forms once it’s inside the body. Hydrogels are water swellable networks, which can be utilized for this purpose. Several approaches can be used for hydrogel gelation, including the use of a thermosensitive molecule; but this can be toxic for the cells, and thus for the patient.
Cell survival in hydrogel

Denitsa Docheva and her team at the University Regensburg Medical Centre, are working to develop a thermosensitive hydrogel associated with TSPCs.
This thermosensitive hydrogel hardens at 25°C and has shown low toxicity for TSPCs, with overall satisfying levels of TSPC survival, proliferation and metabolic activity. The hydrogel is made of butane diisocyanate (BDI) hydrogel and collagen type I. Collagen type I is the main component of the tendon ECM and is responsible for the biocompatibility of this hydrogel.
The hydrogel also demonstrated good stability. Indeed, the thermosensitive hydrogel didn’t change in shape and weight compared with a collagen type I hydrogel used as a control, making this new hydrogel more reliable.
Gene expression
The authors studied genes involved in the tendon ECM, and other tendon-related genes, as they are identifying features of the healing tendon. They observed that biomarkers for tendon were expressed. They also saw that some genes were down-regulated, but this seemed to be compensated by the up-regulation of genes with similar functions. One example is that a gene involved in crosslinking of collagen to form the hydrogel was down-regulated while an enzyme with this function was up-regulated. Understanding how and why, for similar genes, some are down-regulated and others are up-regulated is a highly interesting research subject.
The authors also observed that the thermosensitive hydrogel allowed blood vessel penetration (angiogenesis). This parameter is important as blood vessels supply nutrients, oxygen and immune cells (the immune system is also involved in the regeneration process), and also remove waste.
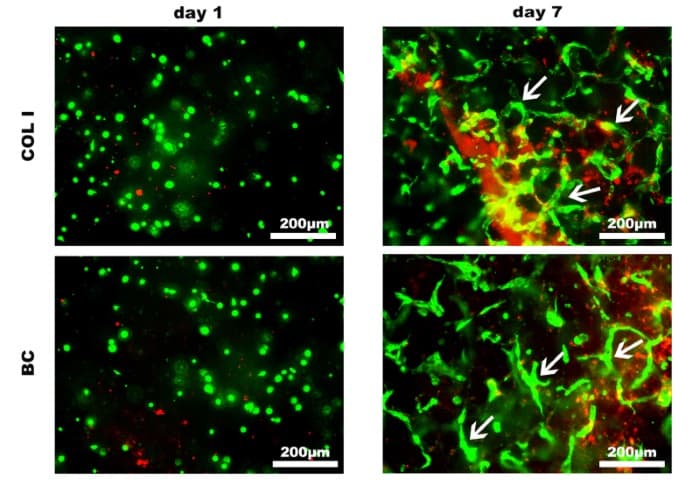
These results are encouraging and the next steps aim to test this hydrogel scaffold in relevant in vivo models, with the aim of recognizing the therapeutic potential of this system in a clinical setting.



