Decellularization of tissues for regenerative medicine applications, such as cartilage repair, is the subject of much research effort. The principle of naturally derived extracellular matrix (ECM)-based tissue engineering is to remove cells from the tissues without affecting their ECM, leaving an intact niche for cell repopulation and, hence, enabling enhanced healing (see: Decellularized and recellularized grafts repair injured cartilage).
Taking this a step further, the decellularization of whole organs offers the possibility of achieving the same goals in a complete structure such as a heart. This would revolutionize the fields of transplantation and regenerative medicine.
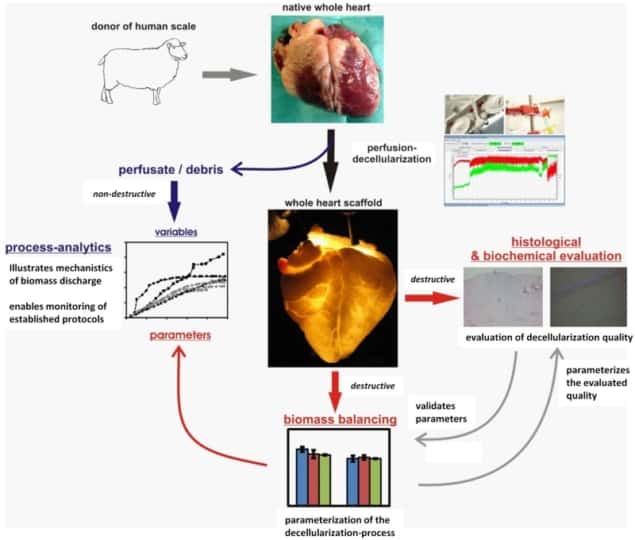
Whole organ decellularization is achieved through perfusion, which delivers the decellularizing agents (detergents and enzymes) using the tissue’s own vasculature. Obviously, this is a complex process and there are many variables that can influence the decellularization and affect the microstructure of the organ. In addition, research in this field has mainly focused on small animals like rats (which have much smaller organs), and is now more and more striving towards human-sized organs. Thus, the field of tissue engineering needs further research to understand the processing of whole organs by perfusion, particularly in large animal models.
Standardizing and optimizing
To shed some light upon the decellularization of whole hearts, Jörn Hülsmann, Hug Aubin and colleagues from the working group of Payam Akhyari and Artur Lichtenberg at Heinrich Heine University Düsseldorf have published a study in which they assess the processing of small and human-scale heart models. They performed decellularization of rat and ovine hearts and analysed the process by-products (perfusates or the solution that runs through the organ) and the effects on both organs. As result, they observed how the process presented varying characteristics that may help further its understanding and optimization (Biomed. Mater. 13 035014).

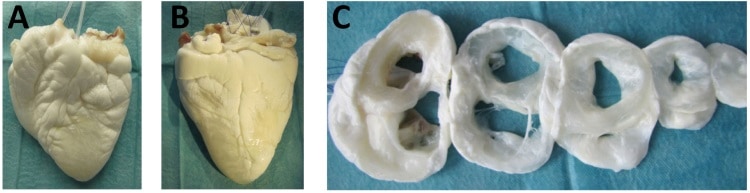
First, they confirmed the efficacy of the decellularization using macroscopic observation, histology and quantification of DNA and cell markers, which demonstrated a high removal of cellular material, while maintaining the tissue ultrastructure and extracellular matrix architecture. Later, the researchers investigated the dynamics of this process via analysis of the by-products or perfusates.
What are we losing?
Ideally, decellularization should only remove the cellular material, which comprises around 19% of the organ’s wet tissue weight. This can be observed after phase separation, drying and weighing of both hearts. Additional exposure to detergents harms the scaffold and, finally, may lead to its de-functionalization.
Surprisingly, after analysing the perfusates, the researchers observed that the detectable protein content was much higher, particularly in rodent hearts, where it represented 71% of depleted mass versus 37% in the larger models.
These differences between small and large organs may be attributed to varying physico-chemical interactions, potentially due to a size effect or to physiological differences (the lipid fraction in ovine hearts is higher, which can affect the action of the decellularizing agents). Furthermore, within the ovine hearts, important differences appeared between individual’s hearts. The researchers related such differences to different discharge dynamics (the speed at which a component is eliminated) produced by effects such protein interactions, debris formation or inherent organ characteristics that complicate the distribution of the solutions. These discordances are evidence for the need to further illuminate and optimize the decellularization of human-like organs.
A more detailed approach
From another point of view, this study presents a more detailed follow-up on the perfusion decellularization of organs. The combination of the analysis of discharged proteins and the viscosity of the decellularizing solutions offers added value that could be implemented into current protocols and improve the control, reproducibility and outcomes.
In future, such a protocol could improve the assessment of the efficacy of the decellularization process and the effects on the organ structure, and help optimize the process during its different phases. Furthermore, it could be a first step towards an automated set-up for production screening or industry production of decellularized whole organs. However, the authors point out that some limitations must still be solved with, for example, the use of more sophisticated and accurate detection systems.



