
Researchers at Rice University in the US have developed a wireless implant that stimulates neurons in response to magnetic pulses. To enable remote operation, the team designed a novel metamaterial that converts external magnetic fields into electrical signals that can be transmitted by nerves. In a study reported in Nature Materials, first author Joshua Chen and colleagues demonstrate that the device can restore nerve function in rats. They propose that this new class of materials could facilitate less invasive medical treatments for neurological and mental health conditions.
Stimulation of the nervous system with electrical fields has been explored to treat a range of disorders including Parkinson’s disease and depression. An electrode is implanted into the brain or nerve, and connected by wires to an external device that sends electrical signals to the target tissue. The aim of this latest research was to build an implant that would operate remotely, requiring less invasive surgery.
“There’s a therapeutic benefit that we’re trying to achieve, without a big device that we put inside the body,” explains neuroengineer Jacob Robinson who led the project.
Such an implant would need to send a signal to cells in response to an external stimulus, and to do this with millisecond lag times, whilst also being small enough to deliver a targeted response. This combination of properties is not found in nature, or in existing engineered materials.
Meeting the requirements of remote neural stimulation
Magnetic fields penetrate deep inside the body but stimulate cells less efficiently than electrical fields. To enable remote cell response, the team designed a magnetoelectric metamaterial that converts an alternating magnetic signal into an electric field. On either side of the device is a layer of material that produces a strain in response to a magnetic field, and at the centre is a piezoelectric material that produces an electric field in response to strain.
Other scientists have studied magnetoelectric materials for remote stimulation of neurons, but these devices have been too slow to mimic neural signalling. To minimize lag times, the material should be driven at its resonant frequency, which is generally a few hundred kilohertz. The membranes of nerve cells, however, filter out high-frequency signals, so previous devices have been driven far from resonance.
The idea the team developed was that the implant could be engineered to stimulate cells whilst driving the material at resonance by converting the current in the device from AC to DC. To achieve this, they deposited a thin film diode on the metamaterial such that the current during AC operation would flow mostly in one direction, resulting in a DC bias.
Implant shows in vivo promise
As a proof-of-concept, the researchers demonstrated that the device could be used to restore nerve function in an animal model. They attached the implant to a severed sciatic nerve in the leg of a rat and showed that applying a magnetic pulse stimulated the muscles in the animal’s foot. The implant achieved a target lag time of 5 ms, which is equivalent to neural communication speeds in the body.
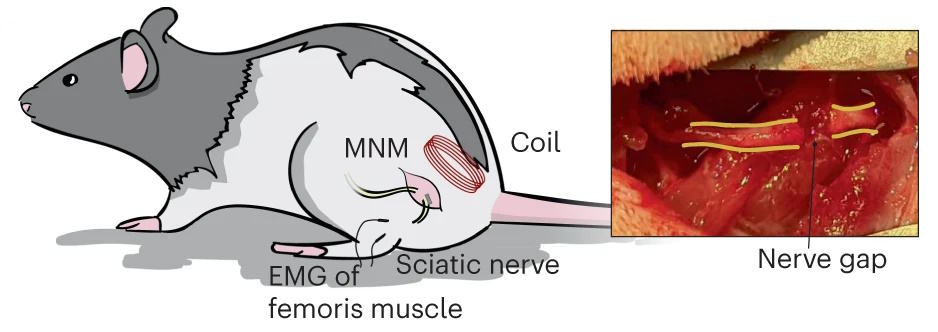
The device accomplishes electromagnetic manipulation that cannot be replicated with naturally occurring materials. The researchers plan to investigate whether the implant can be miniaturized to the micro or nanoscale, enabling it to be used in the brain and potentially in an injectable form.
Biodegradable guide can repair gaps in damaged nerves
Robinson adds that they are also considering other applications, such as power sources, where novel magnetoelectric effects could be harnessed. “There is I think, a whole class of meta materials that we can create, where the relationship between the magnetic field and the electric field is ours to engineer,” he says.




