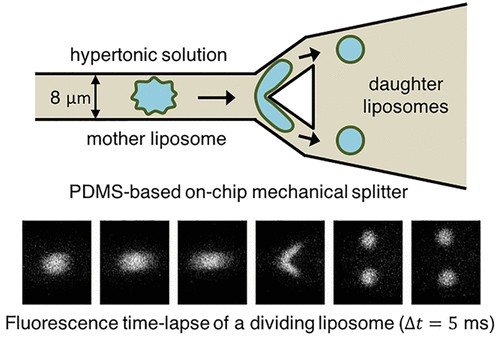
Liposomes, aqueous vesicles with a lipid-bilayer membrane much like our own biological cells, can be split into two stable daughter liposomes using a mechanical splitter. The division process is highly symmetrical, that is, it produces two roughly equally-sized “daughters”, and occurs in just milliseconds. The new technique might not only provide a way to mimic the growth-division cycle of cells, it might also be used to divide liposomes in an exponential manner to make them smaller and smaller, which may be important for drug-delivery applications. It might even help shed more light on how life itself originated on Earth.
Liposomes are routinely employed as objects for both fundamental and applied studies. For example, they can be used as drug-delivery vehicles in medicine. They can also be filled with biomolecules to mimic biological cells.
Cell division, which produces one or more “daughter” cells from a “mother” cell with transfer of genetic material to the daughter(s), is one of the fundamental characteristics of living cells. However, reproducing this process in the lab is no easy task.
Floppy mother liposomes
“We have now demonstrated for the first time that we can mechanically split liposomes into two daughter liposomes,” says Cees Dekker of the Kavli Institute of Nanoscience Delft, who led this research effort. “We did this in a very intuitive way by running them onto a mechanical splitter that sliced the liposomes in two. While this sounds very logical and simple, it required some fine control of the surface-to-volume ratio of the liposomes since we needed to make the mother liposomes very ‘floppy’ so that the daughter liposomes could survive and did not burst.”
Dekker and colleagues made their liposomes using a technique that they recently developed called octanol-assisted liposome assembly (OLA) to produce liposomes on a chip.
Bubble blowing
“This technique is similar to bubble-blowing,” explains team member Siddharth Deshpande, lead author of the study. “As mentioned, we make the liposomes floppy as they are produced so as to have enough excess surface area to make division possible. We then run these liposomes against a Y-shaped splitter located on the same chip and split them symmetrically into two daughters.”
The process produces equal-sized daughter cells and occurs in just milliseconds, he tells nanotechweb.org. “It does not require any sophisticated protein machinery (unlike biological cells) and there is only limited leakage – that is, only a tiny amount of material inside the liposomes is lost to the surroundings.
Applications in synthetic biology
“We also have a good degree of control over the division process – for example, depending upon the liposome size, we can predict whether it will split, burst like a balloon or snake. Bursting often occurs in bigger liposomes and snaking in ones that are too small (they simply pass through the Y-branch).”
From a synthetic biology point of view, the fact that we can simply divide liposomes in a such a brute-force manner is very interesting, he says. “One of our goals is to mimic the growth-division cycle of cells and such division could be one half of the cycle. Such repeated division cycles might also be used to produce ever smaller liposomes, which might be important for drug delivery applications.
Fundamental questions
“What is more, the technique may even help us better understand how life originated on Earth,” he adds. “Might primitive cells have relied on such simple ways of dividing themselves at the very beginning?”
The team, reporting its work in ACS Nano DOI: 10.1021/acsnano.7b08411, will now be trying to couple this division process to the growth of liposomes. “We are currently working on new techniques to grow liposomes and it will be quite an achievement to combine these two modules to produce the complete ‘life cycle’ of these aqueous vesicles.”



