
A nanodiamond-based sensor that measures the thermal conductivity of living cells at subcellular resolution could help researchers better understand how organisms generate and control heat. By combining heat delivery and sensing into a single device, the new nanosensor could also aid the development of heat-based cancer therapies.
All mammals and birds produce their own internal heat. So, too, do certain plants and fish. However, the mechanisms that organisms use to generate and control this heat – which arises from intracellular biochemical sources and then flows through cells to warm up the entire body – are not completely understood.
A technique known as luminescence nanothermometry is often used to measure heat flow in living cells because its resolution is high enough for researchers to pinpoint temperature variations within cells. These variations can be significant, with recent studies indicating that the temperature in one part of a cell may be as much as 1 K warmer or cooler than another.
Measuring local intracellular thermal conductivity
There is, however, a problem with these measurements: the temperature increase observed in laboratory experiments is several orders of magnitude larger than the increase predicted using physics-based models of cellular heat generation. To investigate this disparity, researchers at Osaka University, Japan; the University of Queensland, Australia; and the National University of Singapore created a two-in-one device that acts as a temperature sensor while also generating heat at the same location. This device enabled team members to measure the local intracellular thermal conductivity – an essential parameter of cellular heat-generation models.
The new device, which was made by a team led by Madoka Suzuki of Osaka’s Institute of Protein Research, consists of fluorescent nanodiamonds coated with a heat-releasing polymer called polydopamine (PDA). When irradiated with laser light, the nanodiamonds emit light while the PDA heats up.
The researchers placed their hybrid device inside two types of biological cells, HeLa and MCF-7. They found that the PDA heated up more in cells with a low thermal conductivity than in those with a higher thermal conductivity. This is because heat dissipates more slowly in the former than in the latter. Since the fluorescence of the nanodiamonds depends on their temperature, the researchers were able to use this fluorescence to calculate the rate of heat flow from the device to its surroundings. The technique’s spatial resolution of 200 nm also allowed them to make measurements at different points inside the cells.
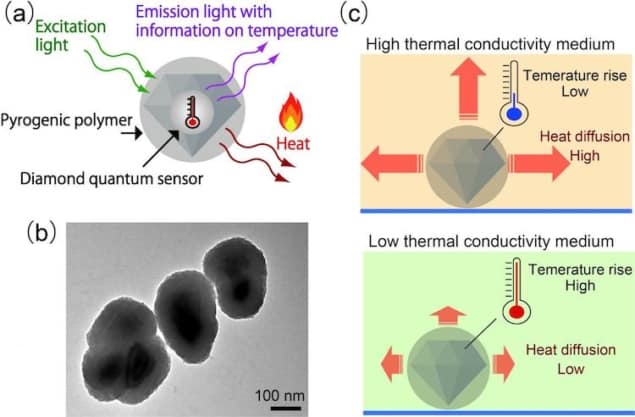
Suzuki and colleagues calculated that the mean thermal conductivity values for the two cell lines were roughly 0.11 W/m/K. Team member Taras Plakhotnik, a physicist at Queensland, describes this as a “fascinating” result because it is much smaller than the mean thermal conductivity of water, which is about 0.6 W/m/K. Since water is an important component of living cells, this result needs a comprehensive theoretical explanation, Plakhotnik says.
Therapeutic uses
As well as a tool for studying cellular conductivity, the researchers believe that their PDAs could also prove useful in heat-based (photothermal) treatments for cancer. Such treatments work by raising the temperature of cancerous cells high enough to kill them, but doing so often kills nearby healthy cells in the process. Understanding heat flow in both types of cell would make the treatments safer, Suzuki says. The heating effect of the PDAs could also be made more efficient by attaching gold nanoparticles to their surface. These nanoparticles are good absorbers of electromagnetic radiation, and heat up thanks to a phenomenon called surface plasmon resonance in which surface electrons play an important role.
Heat transport theory goes universal
Members of the team now plan to improve the accuracy and spatial resolution of their measurements. “We would like to understand why we have observed such a low thermal conductivity for the two cell lines we studied,” Plakhotnik and Suzuki tell Physics World. “Such fundamental work may allow us to explain the origin of ‘hot spots’ observed in biological cells.”
The research is detailed in Science Advances.




