High-grade gliomas such as glioblastoma multiforme (GBM) pose one of the biggest challenges in clinical oncology. The gold standard treatment for these aggressive brain tumours — resection followed by radiotherapy and chemotherapy — is limited by high recurrence rates, therapy resistance and devastating side effects. One potential alternative could lie in proton minibeam radiation therapy (pMBRT).
Minibeam radiotherapy uses an array of parallel, submillimetre-sized radiation beams to deliver spatially fractionated dose. Proton minibeams have already demonstrated a remarkable reduction in neurotoxicity compared with standard proton therapy. Now, a French research collaboration has evaluated the outcomes of pMBRT in glioma-bearing rats (Sci. Reports 10.1038/s41598-018-34796-8).
The team used a passive scattering beamline at the Orsay Proton Therapy Center to irradiate the animals with the plateau region of a 100 MeV proton beam. They note that this energy could be used to treat a tumour located at the centre of the human brain. “The experiments were performed in conditions that can be compared with, or translated directly to, possible human irradiations,” points out first author Yolanda Prezado, from the IMNC laboratory at CNRS.
To generate the minibeams, the researchers used a multi-slit brass collimator with 400-μm-wide slits, spaced 3.2 mm apart and positioned 7 cm from the rat skin. This set-up produced minibeams with a width of 1.1 mm at 1 cm depth in the rats’ brain. Prezado notes that clinical irradiations could use a pencil-beam scanning system with multi-slit collimators adapted to fit the tumour size. “The beam widths and spacing to be used in potential clinical trials are the same as those being used in small-animal experiments,” she explains.
Efficacy and safety
To evaluate the tumour control efficacy of pMBRT, Prezado and colleagues treated nine rats with aggressive RG2 glioma tumours. The animals received one fraction of pMBRT to the whole brain (excluding the olfactory bulb) with peak doses of 70 Gy at 1 cm, corresponding to a mean dose of 30 Gy.
The 3.2 mm spacing between the beams exposes the rat’s brain to a highly heterogeneous proton dose distribution, with large areas of tumour receiving non-lethal radiation doses. Dose–depth profiles revealed peak-to-valley dose ratios of 6.1 at the tumour position and 6.5 in the centre of the brain.
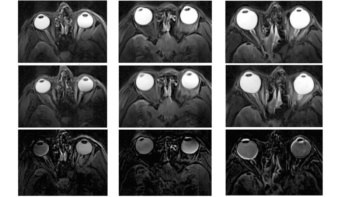
Treatment with pMBRT provided significant tumour control. The mean survival time of the irradiated rats was 32.5 days, compared with 18 days for a control group of seven non-irradiated tumour-bearing rats. Two irradiated animals survived for the entire study duration and were sacrificed six months after irradiation, at which point no tumour was seen in MR images or histopathological analysis. The authors emphasize that tumour eradication was achieved despite delivering a highly heterogeneous dose distribution in just one fraction.
Any rat showing adverse neurological signs related to tumour growth was humanely killed. The non-irradiated tumour-bearing rats displayed large gliomas at the moment of the sacrifice. Irradiated rats sacrificed less than three months after irradiation (due to symptoms of tumour growth) also had large gliomas plus necrotic foci in the tumour and peripheral brain tissue.
Safety check
The researchers also investigated the long-term normal-tissue toxicity of pMBRT, comparing the outcome of nine normal (tumour-free) rats that received pMBRT with a second control group of five non-irradiated normal rats.
The irradiated normal rats gained weight as expected and did not show any external clinical symptoms or skin damage, just a reversible epilation in the minibeam path. Six months after irradiation, the researchers performed MRI on five irradiated normal rats. They found no visible lesions in these animals and no significant differences from the non-irradiated controls. Histopathological analysis performed six months after irradiation revealed only minimal lesions. Four rats that were followed up for one year exhibited a similar profile.
The team concluded that pMBRT provides significant tumour control in RG2 glioma-bearing rats, achieving 22% long-term tumour-free survival. In addition, the 70 Gy proton minibeams did not induce the side effects, such as radionecrosis, that would have resulted from standard proton therapy at lower doses.
Proton minibeams spare critical brain structures
This combination of increased tumour control and reduced neurotoxicity widens the therapeutic window for high-grade gliomas, possibly enabling more aggressive irradiation schemes. The fact that large areas of the brain could be irradiated without significant side effects may overcome one of the major difficulties of treating gliomas: its infiltrative nature. Furthermore, as pMBRT does not require homogeneous target coverage to achieve tumour control, it could reduce positioning and targeting precision requirements.
As for the mechanisms underlying these results, the team suggest that non-targeted effects, such as cell signalling or changes in the immune microenvironment, may play a role. Another possibility may be a preferential effect on the tumoural versus normal vasculature. The team’s next challenge will be to “unravel the biological mechanisms involved,” Prezado tells Physics World.