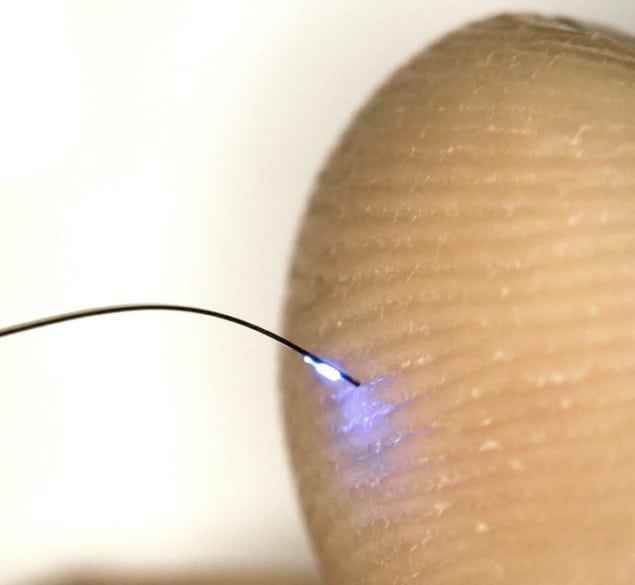
A new wireless optoelectronic photometer comprising an ultrathin flexible injectable probe containing light sources, photodetectors and a miniature power supply could be used to record neural activity in animals as they go about their daily activities. The new device is very different from the cumbersome and movement-restricting fibre-optical-waveguide systems in use today and records fluorescence signals associated with the flux of calcium ions in neurons – for example, in the basolateral amygdala, a deep-brain structure involved in processing emotions.
One of the main goals of neuroscience today is to better understand the connection between neuronal processes and behaviour. Most techniques today to monitor neuronal activity in the brain rely on detecting the fluorescence from genetically-encoded calcium “indicators” (GECIs) in neurons. The problem is that these often rely on using bulky fibre-optical waveguides that are placed on an animal’s head and which prevent it from moving around normally.
A team of researchers led by John Rogers of Northwestern University in the US has now developed an alternative compact photometry system for recording fluorescence signals from deep within the brain. The device contains an ultrathin flexible and lightweight injectable probe containing components for stimulating the brain with light and then recording the fluorescence from the GECIs in neurons. These signals are then sent wirelessly to a receiver, and a detachable transponder, control unit and miniature power supply are also included in the device.
Very thin probe
The technique employed by the researchers is a relatively new one known as optogenetics, which uses light to control and monitor the behaviour of cells in living tissue, usually neurons, that have been genetically modified (using proteins) to become sensitive to light. It has revolutionized neuroscience in recent years, since researchers can now study how neurons behave in real time.
“In our study, we created cellular-scale blue light-emitting diodes and photodetectors and mounted these on thin, filamentary polymer probes that we could insert in the brain,” explains Rogers. “The injectable part of the probe has a maximum thickness of just 150 microns, which minimizes damage to tissue. An ultra-miniaturized wireless control and data communication unit mounted on the head of the animal being studied (primarily mice in our case) allows us to record fluorescence signals correlating to the level of brain activity in a region of interest.”
Mice are less stressed
An essential aspect of the technology is that it is light and there are no trailing electrical wires that can get in the way of the animal. What is more, the devices allow for simultaneous recording from several animals at the same time, which will help in better understanding complex behaviour associated with social interactions, for example.
Rogers and colleagues measured fluorescence signals associated with GECIs in the basolateral amygdala of mice and say that the results they obtained are on a par with those obtained using traditional fibre-optics systems. Calcium-ion flux in neurons is directly related to neuronal activity and the basolateral amygdala is an important region of the brain for processing emotional information. It is especially active in rodents when they are afraid or anxious. As a bonus, the researchers say that the animals in their experiments were less stressed compared to those connected to a traditional fibre-optic system.
Towards more advanced, battery-free systems
“Our wireless system allows us to perform neuroscientific studies that are simply impossible with the type of fibre-optic systems available today,” Rogers tells nanotechweb.org. “Such studies include social interactions and natural movements in realistic 3D environmental enclosures.”
The team, reporting its work in PNAS doi.org/10.1073/pnas.1718721115 says that it is now busy building advanced, battery-free systems that are even smaller and so can be implanted directly under the skin. “We are also integrating other kinds of functionalities, such as electrical sensing and simulation, optogenetics and fluidic delivery, on the same platform,” explains Rogers.
“For instance, adding red LEDs to the injectable probe would allow for simultaneous optogenetic stimulation with redshifted channelrhodopsins (molecules that can be remotely activated with light) and calcium-ion recording from dopaminergic neurons in the ventral tegmental area. This could allow us to study behaviour such as the reward response in animals.”



