The most accurate measurement yet of the half-life of iron-60 has been made by an international team of physicists. While previous measurements of the half-life had differed by a factor of two, the team says that it has accounted for most sources of error that plagued earlier experiments. The radioactive isotope – whose half-life is measured at 2.60 million years with a 2% uncertainty – can now be used to date astrophysical events on that timescale, making it a reliable astrophysical chronometer.
While most of the iron in the universe is iron-56 – a stable nucleus made up of 26 protons and 30 neutrons – iron-60 (60Fe) has 34 neutrons and it is the four extra neutrons that make the isotope unstable to radioactive decay. 60Fe is also an “extinct radionuclide” – a nuclide formed by primordial processes in the early solar system, nearly 4.6 billion years ago.
Solar system histories
The existence of such radionuclides is normally inferred by looking for a “superabundance” of their stable decay products – one neutron becomes a proton and 60Fe decays into cobalt-60 (60Co) and finally into the stable nickel-60 (60Ni). While there is no primordial 60Fe still on the Earth from when the solar system formed, a small amount has been found in the deep-sea of the ocean floor. This material most likely originates from outer space – either from meteorites (where 60Fe is produced thanks to the meteorites being constantly bombarded by cosmic radiation) or from stellar nucleosynthesis events such as supernovae that occurred only a few million years ago. Such traces help astronomers build a clearer picture of the supernovae that may have occurred nearby as the solar system evolved, having a significant impact on the Earth’s climate, as well as its ability to host life.
To use 60Fe as an accurate chronometer though, its half-life must be known precisely. But the two best previous measurements – made in 1984 and in 2009 – found different answers that disagreed by nearly a factor of two. Indeed, the 1984 experiment found the half-life of 60Fe to be 1.5 million years, while the 2009 experiment deemed it to be 2.6 million years.
Perfectly precise?
Anton Wallner from the Australian National University, along with colleagues in Austria and Switzerland, has now confirmed the latter measurement of 2.6 million years with a 2% uncertainty by using a different method from the 2009 group.
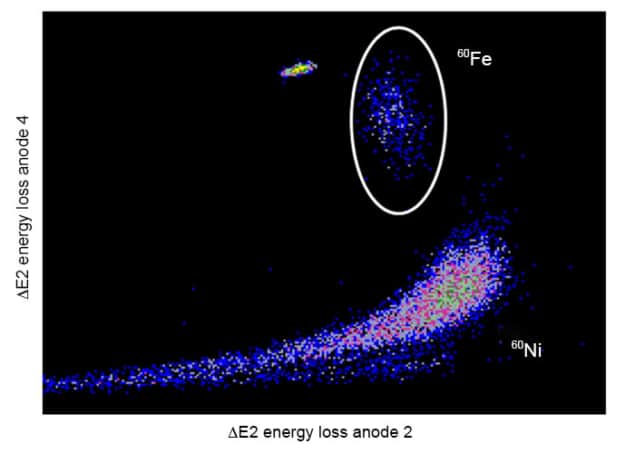
Scientists normally measure half-lives by using different types of spectroscopy to measure the decay rate per second in a sample containing a known number of the nuclei. For 60Fe, they detect the gamma rays that are emitted by its daughter nucleus 60Co, which is no mean feat. Wallner tells physicsworld.com that two factors play a key role – the experiment needs a sufficient number of 60Fe nuclei within its sample, so that the radioactivity is high enough to be measured. Furthermore, the team must know to great precision the initial number of 60Fe nuclei in the sample. Ironically, this number is directly related to the half-life value itself, making the entire measurement very tricky.
Paradoxical measurement
The team used accelerator mass spectrometry (AMS) – the same method used by the 1984 experiment, but different from the 2009 experiment – to determine the tiny concentration of 60Fe isotopes in its sample. Wallner explains that AMS is better than the 2009 group’s method of “inductively coupled plasma mass spectrometry” (ICP-MS), as AMS allows the team to easily distinguish between equal mass isotopes of different elements present in the same sample. Wallner acknowledges that AMS does have a more complex measurement set-up than IC-PMS, allowing systematic errors to creep in, but the researchers can correct for these errors confidently. “Our motivation was to produce an accurate value, even if it is not as precise as the ICP-MS result,” he says. Instead of using the 60Fe half-life, they compared the number of isotopes detected to the concentration of 55Fe – another rare isotope – thereby accounting for the aforementioned errors.
There are numerous implications of having nailed down this value because primordial radionuclides (whose half-lives span between 0.3 and 81 million years) allow astrophysicists to deduce the time-scales of the process that they are produced in. These include supernovae and evolving low-to-medium-mass (AGB) stars. Also, Wallner explains that “60Fe is produced in stellar nucleosynthesis by neutron capture on 59Fe,” but, as the neutron-capture cross sections are directly related to the half-life, knowing this value is essential. There are satellites, such as ESA’s International Gamma-Ray Astrophysics Laboratory (INTEGRAL), that can detect “recently” decaying 26Al and 60Fe in our galaxy by counting the number of atoms present in the interstellar medium. Once again, this requires an accurate half-life value.
- The research is published in Physical Review Letters.



