Nanotechnology could transform solar cells from niche products to devices that provide a significant fraction of the world’s energy, as Edwin Cartlidge discusses

The burning of skin on a hot summer’s day, the awesome power of a tornado or the existence of a simple blade of grass all testify to one thing: the huge amount of energy transmitted to us from the Sun. In a single hour the Sun delivers the same amount of energy as consumed by all of humanity in a year – about 5 × 1020 J – and in 36 hours releases as much energy as exists in the Earth’s estimated oil reserves. When you combine this with the fact that solar energy is essentially inexhaustible, available to everyone the world over and generates no greenhouse gases or other harmful pollutants, it seems hard to imagine why we do not make greater use of it.
The main reason is cost. Electricity produced by solar (or photovoltaic) cells costs about $0.30 per kilowatt hour (kWh), whereas that derived from wind costs in the region of $0.05 per kWh and from natural gas about $0.03 per kWh. Technologically, the difficulty in deriving energy from the Sun’s rays – as opposed to, say, fossil fuels – is that they have a relatively low energy density. The upshot is that in the US, for example, photovoltaic cells generate only about 0.02% of electricity, with the vast majority of the rest coming from coal, gas and nuclear power.
This situation is set to change, however. Gradual improvements to the basic single-crystal silicon solar cells have already reduced the cost of photovoltaic electricity by about a factor of 20 in the past 30 years, and the continuing development of cheaper crystalline materials should see this trend continue. According to a report produced by American scientists George Crabtree and Nathan Lewis for the US Department of Energy in 2005, solar cells will become competitive enough – generating electricity at $0.02 per kWh – to be implemented on a massive scale in about 20–25 years’ time.
But some believe that the rise of solar energy could be far more dramatic. Crabtree and Lewis themselves estimate that the widespread use of photovoltaic cells could happen as soon as 2015 if physicists can perfect a new generation of more advanced devices built using nanotechnology. These include cells based on quantum dots or nanocrystals devices, which are potentially both cheaper and more efficient than existing cells. Indeed, such is the promise of these technologies that a report produced for the German government in 2003 predicted that by 2050 photovoltaics could be meeting a quarter of the world’s energy needs.
Such a transformation would require huge political will. However, the increasing urgency with which governments are addressing, or at least discussing, the issue of climate change suggests that this will may emerge. Although the world’s emissions of greenhouse gases could be reduced using other non fossil-fuel energy sources, some researchers, such as solid-state physicist Keith Barnham of Imperial College London, believe that photovoltaics could take much of the strain on their own. He points out that if the UK expanded its photovoltaic capacity by 40% each year – which is less than occurred globally in 2004 – then it could more than make up for the loss of generating capacity caused by the closure of its ageing nuclear reactors over the next 20 years.
Solar-cell basics
The silicon solar cell has remained essentially unchanged since it was invented at Bell Labs in the US over 50 years ago. Part of a wafer of silicon is doped to create an excess of holes (i.e. a p-type semiconductor) while another part of the wafer is doped to contain an excess of electrons (an n-type semiconductor). At the junction between these two regions, electrons and holes combine to create a potential barrier, which keeps the remaining electrons and holes apart. However, when a photon with sufficient energy strikes the cell, it promotes electrons from the valence band to the conduction band, creating electron–hole pairs. Pairs formed on or near the p–n junction are forced by the electric field to separate so that the holes pass to the p-type region and the electrons to the n-type region, thereby producing a current.
The performance of a solar cell is measured by its efficiency: the ratio of the electrical power generated to the power of the light incident on the cell. In 1961 physicists William Shockley – who shared the 1956 Nobel Prize for Physics for the invention of the transistor – and Hans Queisser calculated that the simplest kind of solar cell can achieve a maximum efficiency of 31%. This is a cell that consists of a single p–n junction, generates just one electron–hole pair for each incoming photon, is exposed to unconcentrated sunlight, and wastes as heat any incoming photon energies in excess of the semiconductor band gap.
The vast majority of solar cells on the market today are so-called first-generation cells, which are made from single crystals of silicon. The best-performing (which is considerably less than the record achieved in the lab, by Martin Green and colleagues at the University of New South Wales in Australia, of 24.7%). However, first-generation cells are expensive to produce because of the high costs of purifying, crystallizing and sawing the single silicon wafer. “Second-generation” solar cells aim to reduce these costs by using thin films of silicon and other, compound, semiconductors, such as copper indium diselenide and cadmium telluride, mounted on glass substrates. But while much cheaper than monocrystalline silicon cells, these second-generation devices suffer from structural defects that make them less efficient than their single-crystal counterparts.
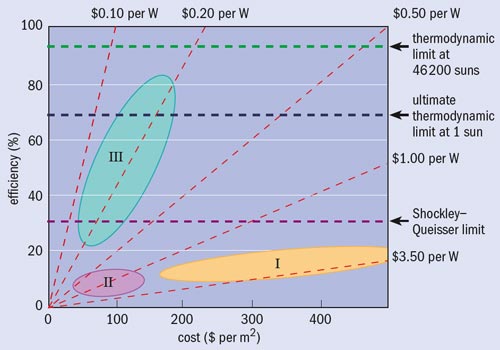
To try and overcome these limitations, researchers are working on third-generation cells that, if practicable, would yield extremely high efficiencies but be as cheap to produce as thin-film devices (see “Generation gap” figure). The way to make these third-generation devices is to violate one or more of the Shockley–Queisser criteria. One option is to concentrate the sunlight using mirrors or lenses. The number of electron–hole pairs, and therefore the current from a cell, is proportional to the rate of photons arriving at the cell. This effect in itself would not yield a higher efficiency since the current per unit flux of sunlight does not increase. But because the output voltage of a p–n junction increases logarithmically with the current, the power output and therefore the efficiency do in fact increase logarithmically. If all other aspects of the cell remain equal, focusing the incoming sunlight can increase efficiencies up to 41%.
Another option is to stack multiple cells with different semiconductor band gaps on top of one another. This arrangement allows the device to generate a current from a much wider range of photon wavelengths than a single-crystal silicon cell. Already used to power spacecraft, where cost is not such a big issue, multi-junction cells have an efficiency limit of 43% if they contain two separate junctions, 49% for three junctions and 66% with an infinite number of junctions.
A further option for exceeding the conventional 31% limit is to convert some of the excess photon energy (i.e. the difference between the photon energy and the semiconductor band gap) into useful energy. One way to do this is to create multiple electron–hole pairs for each incoming photon. For decades it has been known that this phenomenon takes place inside bulk semiconductors, where energetic conduction electrons knock other electrons from the valence band into the conduction band. But this effect is very limited – in silicon, for example, it leads to only just more than one electron per incoming photon.
However, according to physicist Victor Klimov at the Los Alamos National Laboratory in New Mexico, this so-called carrier multiplication can be enhanced by making solar cells from networks of billions of tiny pieces of semiconductor known as quantum dots, rather than one large piece of semiconductor. In experiments performed last year, Klimov was able to generate up to seven electron–hole pairs per incoming photon by illuminating single 5 nm-sized crystals of lead selenide with extremely brief laser pulses. He says that this process could lead to solar cells with efficiencies of over 40%.
Klimov admits that he does not know exactly how the quantum dots manage to achieve this photon multiplication, but he believes it may be due in part to the process that occurs in bulk semiconductors and also, possibly, because of the creation of “virtual electrons”. This latter process would involve an electron gaining more energy than was deposited from an incoming photon, albeit for a very brief period of time, and then transferring some of its excess energy to an electron in the valence band.
Quantum dots could also be used to make “hot-carrier” cells, in which the extra energy supplied by a photon is not lost as heat – as it is in conventional solar cells – but instead results in higher-energy electrons and therefore a higher voltage than in a standard cell. Green at the University of New South Wales is one physicist working on this technology. “When building a material from the bottom up in the form of quantum dots, it is possible to manipulate the properties of the material on the micro-scale,” he explains. “So by changing the stiffness of the interaction between the quantum dots and the material that they are embedded in, we can reduce the amount of heat that is lost due to atomic vibration.”
Although it could be 10–15 years before this technology reaches the market, Green’s group has now built its first cell based on quantum dots, and he thinks that commercial devices based on this technology could reach efficiencies of 20–30%. “I would be very surprised if in 30 years’ time solar cells were not using nanotechnology in some way,” he adds.
The quantum-well cell
Before quantum dots can be used to make solar cells based on either carrier multiplication or hot carriers, however, two crucial hurdles must be overcome. One is how to separate out the electrons and holes generated in such devices – i.e. the function performed by the p–n junction in a silicon solar cell – while the other is to find a way to connect the individual quantum dots. This could perhaps be done by using nanowires or by simply placing the dots close to one another and relying on quantum tunnelling.
An alternative third-generation device that also takes advantage of nano-scale structures is the so-called quantum-well solar cell. Barnham and colleagues at Imperial College London have built such a cell by sandwiching 50 slices of the semiconductor indium gallium arsenide, which has a relatively low band gap, with each slice just a few nanometres thick, between slightly thicker pieces of gallium-arsenide phosphide, which has a higher band gap. Each slice of lower-band-gap material bounded on either side by the higher-bandgap substrate forms a potential well, into which photons are absorbed and from which electrons and holes then escape thanks to their thermal energy.
The quantum wells shift the energy range over which the cell operates downwards, and in doing so reduce the band gap of the gallium arsenide so that it can capture a greater fraction of the spectrum of photons striking the cell. This slightly decreases the voltage from the cell (since the average energy of the electron– hole pairs produced is lower), but this drop in voltage is more than compensated for by the increased current, resulting in a higher power output overall. A more obvious way to reduce the band gap of a gallium-arsenide cell is simply to grow a layer of indium gallium arsenide on top of it. But according to Barnham, mismatches in the atomic spacing between the two materials introduce dislocations that reduce the efficiency of the compound material.
Like other third-generation solar cells, the quantum-well cell would be exposed to concentrated sunlight. Relatively cheap optics focus the incoming light onto a much smaller area of solar cell, thereby lowering the area of cell needed and the overall cost of the system. Since the cell itself makes up a lower fraction of the overall system costs it makes sense to use a more expensive, but higher efficiency, cell. Barnham and coworkers have so far recorded efficiencies of up to 27% in their cell when it was exposed to sunlight that has been concentrated 300 times. The researchers claim that the efficiency of their cell could in fact go beyond 30% by recycling the photons when the electrons and holes recombine, as they inevitably do. Barnham and colleagues recently set up a company, QuantaSol, to commercialize their technology, and plan to sell their quantum-well cells to concentrator manufacturers within the next six to nine months.
Dyes and plastics
An entirely different type of third-generation device is the “dye-sensitized” solar cell. Pioneered by Michael Grätzel of the Swiss Federal Institute of Technology and co-workers, it uses a combination of a chemical dye and the wide-band-gap semiconductor titanium dioxide, which is cheaper than silicon. Photons arriving at the cell liberate electrons from the dye molecule, which are then transferred to the conduction band of the semiconductor and out to an electrode. The hole left in the dye, meanwhile, recombines with an electron in a layer of electrolyte that sits between the semiconductor and a second electrode.

One of the virtues of the dye-based solar cell is that the band gap of the semiconductor does not have to be matched to the spectrum of light impinging on the cell; the absorption spectrum of the dye can be easily tuned to this – which is why the cheap semiconductor titanium dioxide, with its wide band gap, can be used. As the semiconductor layer need not be thick, the dye cell can also be mounted on flexible substrates. In addition, because the cells are transparent they can be embedded into windows.
According to Grätzel, scientists had abandoned this concept because of the limited intensity of light gathered by the dye. But he and his co-workers have shown that this problem can be overcome by using a nano-crystalline form of titanium dioxide. A network of nanometre-sized semiconductor crystals provides a multitude of nooks and crannies in which the dye molecules can bond, multiplying the surface area available to the dye by over 1000 times. Grätzel says that his group’s best-performing cell to date has an efficiency of just over 11%, which should, he estimates, make commercially manufactured dye-sensitized cells between three and four times cheaper than conventional silicon cells. This year the company G24 Innovations started the first commercial production of dye-sensitized solar cells at a plant in Cardiff in the UK.
Finally, a much younger technology than dye-sensitized cells, but one with considerable potential, is the organic solar cell. Such devices, which use plastics as the active component, are potentially far cheaper to make than semiconductor devices. They are also flexible, which means they could be wrapped around surfaces, rolled up or perhaps painted onto structures. Earlier this year David Carroll, a physicist at Wake Forest University in North Carolina, and colleagues claimed to have created a polymer-based solar cell with an efficiency of 6%. Although low by the standards of silicon, this efficiency is nevertheless impressive for polymers, which have a high band gap and are not as good at separating out electron–hole pairs.

Carroll and colleagues achieved their high efficiency by creating nanometre-sized “veins” within the polymer poly(3-hexylthiophene) that guided the electrons and holes rapidly out of the device before they could recombine. However, the researchers believe that they can attain efficiencies as high as 10% by wrapping a polymer around a piece of fibre optic cable. The fibre has two roles: holding the photons within the polymer until they are absorbed; and capturing photons from a greater range of incident angles. This latter point increases the fraction of the day that a cell can perform at peak levels – from about an hour with a thin-film cell to about five hours.
“Plastic solar cells are definitely here to stay,” says Carroll, whose team is currently building prototypes of both the thin-film and fibre varieties. “They make very good flexible cells that are capable of providing more power over the course of a day than silicon. Even six months ago I would not have said this.”
Overcoming the storage problem
Building efficient, and therefore cheap, photovoltaic cells is not, however, a guarantee that solar power will become a major part of the world’s energy mix. Even if these devices can be converted into high-performance commercial products, there still remains the problem of actually building and installing the enormous number of panels that would be needed. Humans currently consume energy at a rate of 13 terawatts (TW), and many experts predict that population growth and economic expansion will increase this figure to about 45 TW by 2050. Generating 20 TW of that energy with panels that are 10% efficient would, according to Crabtree and Lewis, mean installing such panels over 0.16% of the Earth’s land surface. Given that only a fraction of this will be met by installing panels on people’s homes, vast “farms” will have to be built in areas with significant amounts of sunshine. Attempting to build such farms in the West could, ironically, be opposed on environmental grounds.
Another hurdle is the infrastructure needed to deliver the solar electricity to where it is needed (when the cells are built in farms). Perhaps the biggest challenge, however, is how to store solar electricity, given that the Sun does not shine all the time. One option, which is already used by producers of nuclear power, involves pumping water up hill when the demand for energy is low and then releasing the water when demand is high thereby generating electricity in the process. Solar energy could also conceivably be stored using batteries or flywheels (see “Energy storage takes off” by Bob Swarup on page 42, print version only) or even through the creation of hydrogen. However, the infrastructure needed to pump the hydrogen to where it is needed would be extremely expensive.
It therefore remains to be seen whether the current rapid growth in photovoltaic capacity can be maintained. In particular, it is uncertain to what extent governments will back solar power. Germany has set up a programme that guarantees in law that generators of solar energy will be paid a certain minimum amount for their electricity by companies operating electrical grids, which share the extra cost that they incur between their customers. This has resulted in a number of large photovoltaic companies, including Q-cells based near Leipzig in Germany, which is now valued at several billion dollars and is growing extremely quickly. Meanwhile, Japan – which is home to the world’s largest photovoltaic manufacturer, Sharp – has plans to increase its solar-cell capacity to about 100 gigawatts (about 30 times the current global capacity) by 2030.
But other countries do not appear as enthusiastic. In the US, for example, money for photovoltaic research is still hard to come by. The US Department of Energy currently spends about $100m a year on developing solar energy, but only a small fraction of this goes on research into novel technologies. “There are no technical challenges that cannot be overcome,” says Carroll. “That is, there are no physical laws that prevent really high-efficiency devices from being built. The technology will be commercialized if the public sector invests.”
Like others, Carroll believes that solar power will account for a significant fraction of world energy production in 10 to 20 years’ time. But he thinks that this could happen much sooner – in as little five years – if politicians were willing to fund more research. “You can solve anything with science,” he adds. “But you have to pay scientists to do it.”



