Gaucher disease is the most prevalent genetic condition affecting the storage of lysosomes – the “stomachs” of cell biology, which break down a range of biomolecules while secreting substances and playing a role in energy metabolism and membrane repair. Current approaches to treat Gaucher disease hinge on enzyme replacement therapy to counter the deficient activity of lysosomal acid-β-glucocerebrosidase (GCase) at the root of the disease. However, in specific cases affecting the neural system, where symptoms can be particularly aggressive limiting life expectancy to just one or two years, enzyme delivery is hampered by the blood brain barrier.
“Mia Horowitz – who is a world expert in Gaucher disease – asked us if we could find a way to bring the enzyme to therapeutic targets for the disease,” explains Dan Peer, Director of the Laboratory of Precision NanoMedicine at Tel Aviv University in Israel alongside Horowitz. “So the question was can we package β-glucocerebrosidase to deliver it to the brain?”
The challenge was further complicated because no adequate animal models exist for studying the disease. In addition, at the outset of the investigation both the target cells for therapeutic GCase and the necessary dose were unknown. Calling on their knowledge of lipid nanoparticles as potential drug carriers, and the brain cell types with functions likely to facilitate a response to treatment, the researchers combined experiments on mice in vivo and on patient samples ex vivo to demonstrate not just delivery of the enzyme, but therapeutic activity as well.
With a number of unknowns still in the mix, Peer is reluctant to suggest the results prove this approach to treatment “works”. “We have established the tool box with some infrastructure studies in order to understand it better – this is the first milestone, and now we should take it to the next level.”
Putting the pieces together
Speaking to a number of physicians failed to reveal a definite target molecule for Gaucher disease enzyme replacement treatment. Instead Peer, Horowitz and colleagues at Tel Aviv University deduced that as macrophages are the target for treating Gaucher disease in the body, a good bet would be to target microglia – the counterparts for macrophages in the brain. The next challenge was getting past the blood brain barrier, and here reports already existed suggesting lipid nanoparticles might help.
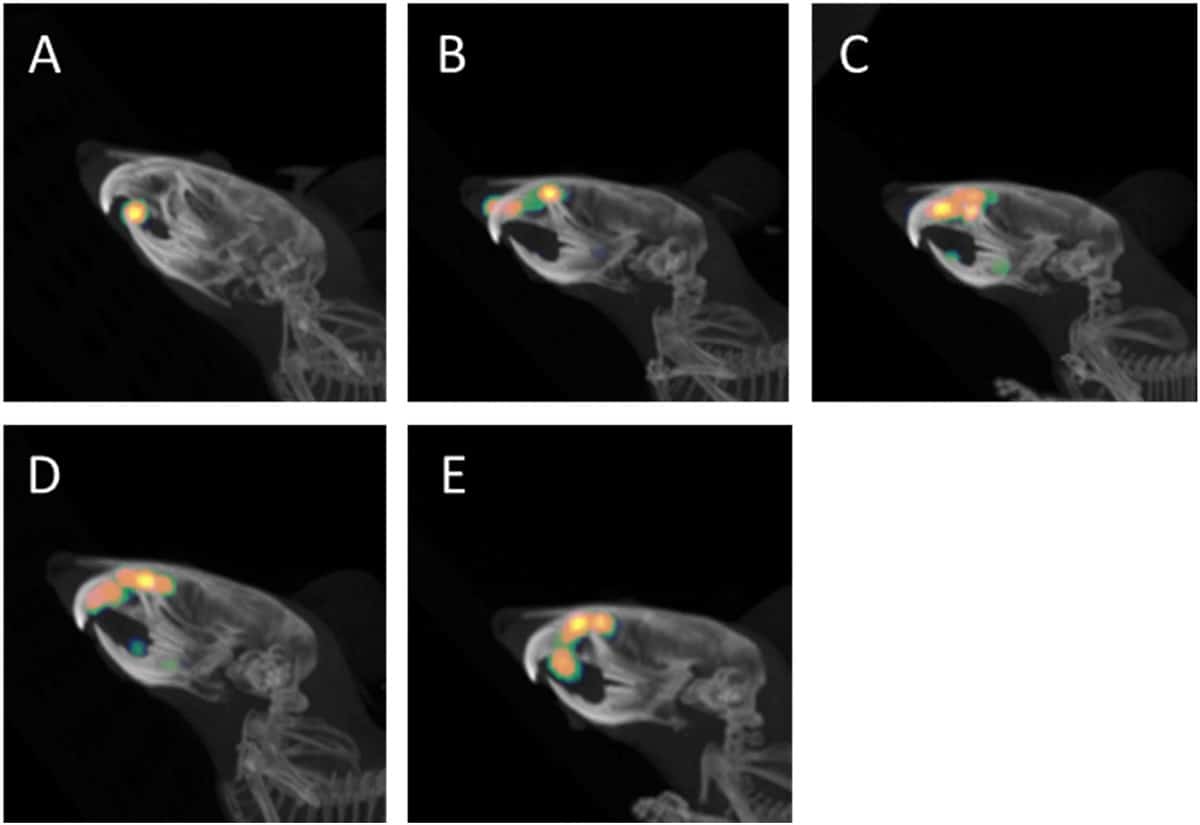
“First we thought can we visualise it?” says Peer, as he describes initial experiments to see whether a fluorescent-labelled protein could reach the brain. Since no animal models accurately recapitulate the disease, the researchers administered the drugs intranasally to live mice to determine whether they could get the enzymes to the target cells in the brain.
With no packaging only 0.3% of the free enzyme reached the brain. When packaged in lipid nanoparticles, crucially incorporating cationic phospholipids (DOTMA), 3.91% reached target – an increase by a factor of ten. Experiments ex vivo were even more encouraging with a 35% increase in enzyme activity in response to the drug delivered in the DOTMA incorporating lipid nanoparticle.
Physical versus chemical paradigms play off in nanomedicine
When is enough enough?
Despite the large improvements with the DOTMA incorporating lipid nanoparticle, Peer highlights that there is still only 3% of the drug getting to the brain. “Is it enough? we don’t know,” he adds. While 3% sounds low Peer also points out that for cystic fibrosis replacing 6% of the target protein is known to be enough for a therapeutic result. “For Gaucher disease, the target cells are supportive cells so they have a therapeutic effect, and 3% might be enough.”
One in every hundred babies born worldwide is a carrier of Gaucher disease, with around one in 20,000 presenting symptoms. In addition links have been suggested between people carrying the disease without symptoms and later development of Alzheimers and Parkinsons. Among Ashkenazi Jewish populations the number of carriers increases to one in every fifteen, making the disease much more prevalent in Israel.
For those born with Gaucher disease the only current treatment is subcutaneous injections of the drug every two weeks for the rest of their life. For those with the more aggressive neuronopathic form of the disease the current outlook is particularly bleak. Despite the remaining unknowns, the first quantitative assessment of using lipid nanoparticles as potential drug carriers for effective intranasally administered treatment of neuronopathic Gaucher disease looks promising.
Full details are reported in Nano Futures.




