Physics involves a lot of chemistry and vice versa at the nanoscale. Bringing nanomedicine from the lab to the bedside relies on getting the right approach for the right circumstances, and whether the mechanism behind the nanotechnology’s activity is physical or chemical can make the world of difference as work by several speakers at Nanotech France 2018 highlighted

Packaging requires specificity
Patrick Couvreur, professor at UMR CNRS 8612 in Paris, France, was publishing work on nanoparticle drug carriers as far back as 1979. His early work in the field included development of biodegradable polyalkylcyanoacrylate (PACA) nanoparticles that could make doxorubicin (DOX) – a widely used anticancer drug – invisible in vivo, helping it get past the multiple-drug-resistant defences of hepatocarcinoma. Further developments in this technology led to the release of Livatag®, which showed so much promise it was fast-tracked through Food and Drug Administration (FDA) clearance. Speaking to attendees of Nanotech 2018, Couvreur pointed out that in some ways Livatag has been a victim of its own success. In phase II clinical trials the Livatag survival curve was better than the “chemoembolization” control arm, where anti-cancer drugs are injected directly into the blood vessel feeding a cancerous tumor. However in the multicentric (conducted at more than one medical centre) phase III clinical trial, the survival curve overlapped the control arm of “polychemotherapy” treatment involving several different drugs.
Couvreur has gone on to develop other medicines based on the same approach, such as monochrome antibody streptavidin for targeting alzheimers. Despite these successes and the time elapsed since the first demonstration of PACA encapsulated DOX, Couvreur told attendees that the number of nanomedicines on the market or even in phase III clinical trials remains very low. He suggested that reasons for this include issues with loading and the fact that a fraction of the drug remains at the surface of the nanoparticles where they are not protected and their release is not controlled. “What was needed was a move from a physical to a chemical encapsulation paradigm using linkers,” said Couvreur.
Chemistry takes over
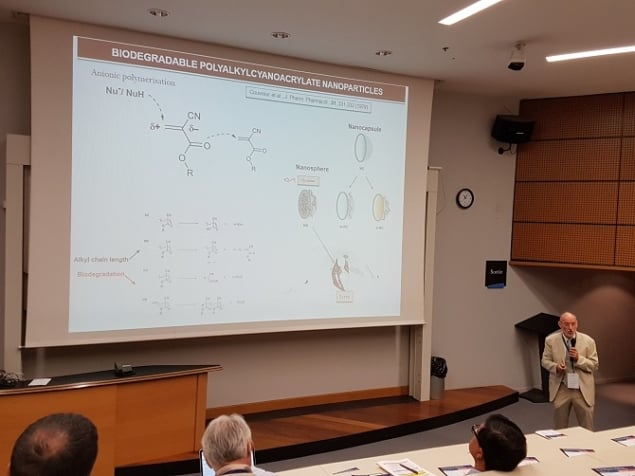
Couvreur described “squalenoylation” as “a new platform for nanomedicines”, giving as an example the success of the nanoassembly of anticancer drug gemcitabine with squalene linkers – SQGem. In the case of doxorubicin linked to squalene nanoparticles, features that improve the medicine’s efficacy include the extension of the nanoparticle by blood flow along streams, which gives longer activity post injection. In addition, interactions between SQGem and lipoproteins mean that it is readily transported by them, particularly cholesterol-rich lipoproteins. Tumour cells attract cholesterol to multiply, so this transport provides an indirect targeting mechanism.
Other applications of squalenoylation include nanoparticles of squalene with cis-diamminedichloroplatinum (CDDP) to increase intracellular delivery of platin and ROS production. Couvreur and his team have also investigated the possibility of combining with adenosine to treat spinal cord injury and brain ischemia. Here the blood-brain barrier can pose a challenge, but Couvreur and colleagues found that the nanoparticles interact with peripheral receptors of adenosine, relaxing the brain vessels and inducing neuroprotection of the brain microcirculation. As a result reperfusion improves while the nanoparticles themselves do not cross the blood-brain barrier.
Getting physical with viruses
Antibiotics – which revolutionized medicine in the 20th century – are among the most frequently administered drugs available on account of their broad efficacy against a range of infectious bacterial diseases. However no such drug exists to combat viral infections – yet. Following on from Couvreur, Francesco Stellacci a professor at Ecole Polytechnique de Lausanne (EPFL) in Switzerland, told attendees at Nanotech France 2018 about work using decorated gold nanoparticles that seem to mimic host cells. The nanoparticles lure the virus in to bond and attempt to infect them, but instead the nanoparticle applies a physical pressure on the virus that results in its rupture and disarmament.
This mechanism here is physical, which has the advantage that it is also non-toxic. “There are a lot of virucidal molecules out there, but they are toxic,” said Stellacci, highlighting alcohol as an example. Mild administration of alcohol may have other benefits but its intake is not effective as a viricide.
The gold nanoparticle viricides also work at nanomolar concentrations – significant since most FDA approved drugs are nanomolar. In addition Stellacci and his team tested them against wild-type viruses extracted from patients and found they are not only effective but that the effect is non-reversible. This means that once the gold nanoparticle viricides have reduced viral populations in a sample by a log 2 difference, the population depletion holds even following dilution of the whole sample, something that is not true of some alternative antiviral substances such as heparin.
Stellacci and colleagues demonstrated the approach on respiratory syncytial virus (RSV), which kills half a million each year. They have also demonstrated the efficacy of the puncturing ligands without the gold nanoparticle, attaching them to cyclodextrin instead. His team are in the process of attempts to apply the approach to combat rotavirus, which leads to diarrhoea, one of the main causes of death in children under five across the world.
Catalysis plays a role
Developments in catalysis may also have spin-off benefits for drug delivery. Jean-Pierre Mahy, professor at the Université de Paris Sud in France spoke to Nanotech France attendees about some of the progress in designing artificial enzymes that “combine the robustness of chemical catalysts with the activity of enzymes in mild conditions.”
Nanotechnology takes on microbial drug resistance
One of the primary goals of his research has been to mimic the cytochromes P450 hemoproteins for selective oxidation – no small feat. The heme moiety of P450s has been described as “responsible for the remarkable and often exquisite, catalytic prowess of these enzymes”. As natural approaches involve multiple electron transfer processes and are very fiddly to reproduce, Mahy and colleagues turned to artificial hemoproteins (hemoartzymes) with mono oxygenase activity. To provide robust structure, high loading, enzyme protection and potential recycling, their recent work has focused on metallorganic frameworks (MOF).
Mahy and colleagues have shown that microperoxidase 8 (MP8) – a heme octapeptide obtained by hydrolytic digestion of horse heart cytochrome c – can have both peroxidase-like and cytochrome P450-like activities. They combined this with an MOF from MIL 101 nanoparticles (where MIL stands for Material of Institut Lavoisier) and were able to demonstrate charge-selective oxidation activity. They have also designed an artificial reductase based on a water-soluble polyimine polymer decorated with hydrophobic groups that allows use of O2 as an oxidant. “This is the first entirely synthetic heme monooxygenase,” said Mahy.
Alongside these demonstrations of the bioactivity of metalloenzymes, the ability to compartmentalize them in MOFs has suggested potential compatibility with living cells, and it is here that possible therapeutic applications really come into play. Some of Mahy’s most recent work has demonstrated artzymes catalysing organic reactions at the surface of living cells. The cells can then enantioselectively catalyse the abiotic Diels-Alder cycloaddition reaction of cyclopentadiene and azachalcone, and as Mahy told attendees, “This could be used to activate drugs.” In addition there is potential for on site synthesis of drugs and metabolites.
Nanotech France is an annual conference that took place this year in Paris on 27–29 June.




